Novel Combinatorial MicroRNA-Binding Sites in AAV Vectors Synergistically Diminish Antigen Presentation and Transgene Immunity for Efficient and Stable Transduction
- PMID: 33995418
- PMCID: PMC8113644
- DOI: 10.3389/fimmu.2021.674242
Novel Combinatorial MicroRNA-Binding Sites in AAV Vectors Synergistically Diminish Antigen Presentation and Transgene Immunity for Efficient and Stable Transduction
Abstract
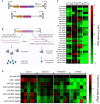
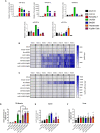
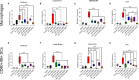
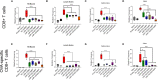
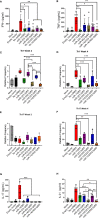
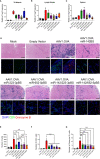
Recombinant adeno-associated virus (rAAV) platforms hold promise for in vivo gene therapy but are undermined by the undesirable transduction of antigen presenting cells (APCs), which in turn can trigger host immunity towards rAAV-expressed transgene products. In light of recent adverse events in patients receiving high systemic AAV vector doses that were speculated to be related to host immune responses, development of strategies to mute innate and adaptive immunity is imperative. The use of miRNA binding sites (miR-BSs) to confer endogenous miRNA-mediated regulation to detarget transgene expression from APCs has shown promise for reducing transgene immunity. Studies have shown that designing miR-142BSs into rAAV1 vectors were able to repress costimulatory signals in dendritic cells (DCs), blunt the cytotoxic T cell response, and attenuate clearance of transduced muscle cells in mice to allow sustained transgene expression in myofibers with negligible anti-transgene IgG production. In this study, we screened individual and combinatorial miR-BS designs against 26 miRNAs that are abundantly expressed in APCs, but not in skeletal muscle. The highly immunogenic ovalbumin (OVA) transgene was used as a proxy for foreign antigens. In vitro screening in myoblasts, mouse DCs, and macrophages revealed that the combination of miR-142BS and miR-652-5pBS strongly mutes transgene expression in APCs but maintains high myoblast and myocyte expression. Importantly, rAAV1 vectors carrying this novel miR-142/652-5pBS cassette achieve higher transgene levels following intramuscular injections in mice than previous detargeting designs. The cassette strongly inhibits cytotoxic CTL activation and suppresses the Th17 response in vivo. Our approach, thus, advances the efficiency of miRNA-mediated detargeting to achieve synergistic reduction of transgene-specific immune responses and the development of safe and efficient delivery vehicles for gene therapy.
Keywords: adeno-associated virus vectors; antigen presenting cells; gene therapy; miR-142; miR-223-3p; miR-652-5p; miR-BS; microRNA.
Copyright © 2021 Muhuri, Zhan, Maeda, Li, Lotun, Chen, Sylvia, Dasgupta, Arjomandnejad, Nixon, Keeler, Manokaran, He, Su, Tai and Gao.
Conflict of interest statement
GG is a scientific co-founder of Voyager Therapeutics, Adrenas Therapeutics, and Aspa Therapeutics, and holds equity in these companies. GG is an inventor on patents with potential royalties licensed to Voyager Therapeutics, Aspa Therapeutics, and other biopharmaceutical companies. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Circumventing cellular immunity by miR142-mediated regulation sufficiently supports rAAV-delivered OVA expression without activating humoral immunity.JCI Insight. 2019 May 21;5(13):e99052. doi: 10.1172/jci.insight.99052. JCI Insight. 2019. PMID: 31112525 Free PMC article.
-
Effective Reduction of Transgene-Specific Immune Response With rAAV Vectors Co-Expressing miRNA-UL112-5p or ERAP1 shRNA.J Cell Mol Med. 2025 Jan;29(2):e70308. doi: 10.1111/jcmm.70308. J Cell Mol Med. 2025. PMID: 39823241 Free PMC article.
-
Combination of miRNA148a-3p binding sites and C5-12 promoter in rAAV vector synergistically reduces antigen presentation and transgene immunity.Life Sci. 2025 Sep 1;376:123742. doi: 10.1016/j.lfs.2025.123742. Epub 2025 May 20. Life Sci. 2025. PMID: 40404114
-
Immune responses to adeno-associated virus vectors.Curr Gene Ther. 2005 Jun;5(3):323-31. doi: 10.2174/1566523054065039. Curr Gene Ther. 2005. PMID: 15975009 Review.
-
Controlled release strategies for rAAV-mediated gene delivery.Acta Biomater. 2016 Jan;29:1-10. doi: 10.1016/j.actbio.2015.10.015. Epub 2015 Oct 22. Acta Biomater. 2016. PMID: 26472612 Review.
Cited by
-
Sub-genomic flaviviral RNA elements increase the stability and abundance of recombinant AAV vector transcripts.J Virol. 2024 Aug 20;98(8):e0009524. doi: 10.1128/jvi.00095-24. Epub 2024 Jul 31. J Virol. 2024. PMID: 39082815 Free PMC article.
-
Recombinant Adeno-Associated Virus Vectors for Gene Therapy of the Central Nervous System: Delivery Routes and Clinical Aspects.Biomedicines. 2024 Jul 9;12(7):1523. doi: 10.3390/biomedicines12071523. Biomedicines. 2024. PMID: 39062095 Free PMC article. Review.
-
AAV Vector-Mediated Antibody Delivery (A-MAD) in the Central Nervous System.Front Neurol. 2022 Apr 12;13:870799. doi: 10.3389/fneur.2022.870799. eCollection 2022. Front Neurol. 2022. PMID: 35493843 Free PMC article. Review.
-
Redundancy in Innate Immune Pathways That Promote CD8+ T-Cell Responses in AAV1 Muscle Gene Transfer.Viruses. 2024 Sep 24;16(10):1507. doi: 10.3390/v16101507. Viruses. 2024. PMID: 39459842 Free PMC article.
-
Gene-based therapeutics for rare genetic neurodevelopmental psychiatric disorders.Mol Ther. 2022 Jul 6;30(7):2416-2428. doi: 10.1016/j.ymthe.2022.05.014. Epub 2022 May 17. Mol Ther. 2022. PMID: 35585789 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

