Alternative Splicing Dynamics of the Hypothalamus-Pituitary-Ovary Axis During Pubertal Transition in Gilts
- PMID: 33995469
- PMCID: PMC8120244
- DOI: 10.3389/fgene.2021.592669
Alternative Splicing Dynamics of the Hypothalamus-Pituitary-Ovary Axis During Pubertal Transition in Gilts
Abstract
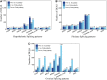
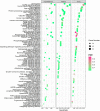
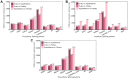
The timing of puberty in mammals marks the point at which reproduction becomes possible. Abnormalities in the timing of puberty may exert a series of negative effects on subsequent health outcomes. Alternative splicing (AS) has not only emerged as a significant factor in the transcription of genes but it is also reported to play a role in the timing of puberty. However, to date, the changes and dynamics of AS during the onset of puberty is extremely seldom explored. In the present study, we used gilts as a research model to investigated the dynamics of AS and differentially expressed AS (DEAS) events within the hypothalamus-pituitary-ovary (HPO) axis across pre-, in-, and post-puberty. We detected 3,390, 6,098, and 9,085 DEAS events in the hypothalamus, pituitary, and ovary when compared across pre-, in-, and post-pubertal stages, respectively. Within the entire HPO axis, we also identified 22,889, 22,857, and 21,055 DEAS events in the pre-, in-, and post-pubertal stages, respectively. Further analysis revealed that the differentially spliced genes (DSGs) associated with staged DEAS events were likely to be enriched in the oxytocin signaling pathway, thyroid hormone signaling pathway, GnRH signaling pathway, and oocyte meiosis signaling pathway. The DSGs associated with DEAS events across the entire HPO axis were enriched in endocytosis signaling pathway, the MAPK signaling pathway, and the Rap1 signaling pathway. Moreover. the ASs of TAC1, TACR3, CYP19A1, ESR1, ESRRA, and FSHR were likely to regulate the functions of the certain HPO tissues during the onset of puberty. Collectively, the AS dynamics and DEAS events were comprehensively profiled in hypothalamus, pituitary, and ovary across the pre-, in-, and post-pubertal stages in pigs. These findings may enhance our knowledge of how puberty is regulated by AS and shed new light on the molecular mechanisms underlying the timing of puberty in mammals.
Keywords: alternative splicing; differentially spliced genes; gilts; hypothalamus-pituitary-ovary axis; puberty.
Copyright © 2021 Pan, Li, Chen, Gong, Li, Jiang, Zhang, Chen and Yuan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- Aliberti P., Sethi R., Belgorosky A., Chandran U. R., Plant T. M., Walker W. H. (2019). Gonadotrophin-mediated miRNA expression in testis at onset of puberty in rhesus monkey: predictions on regulation of thyroid hormone activity and DLK1-DIO3 locus. Mol. Hum. Reprod. 25 124–136. 10.1093/molehr/gay054 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

