Syndecan-1 downregulates syndecan-4 expression by suppressing the ERK1/2 and p38 MAPK signaling pathways in cultured vascular endothelial cells
- PMID: 33997316
- PMCID: PMC8099740
- DOI: 10.1016/j.bbrep.2021.101001
Syndecan-1 downregulates syndecan-4 expression by suppressing the ERK1/2 and p38 MAPK signaling pathways in cultured vascular endothelial cells
Abstract
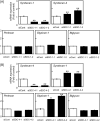
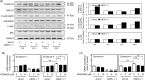
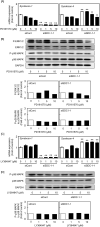
Syndecan-1 and syndecan-4 are members of the syndecan family of transmembrane heparan sulfate proteoglycans. Vascular endothelial cells synthesize both species of proteoglycans and use them to regulate the blood coagulation-fibrinolytic system and their proliferation via their heparin-like activity and FGF-2 binding activity, respectively. However, little is known about the crosstalk between the expressions of the proteoglycan species. Previously, we reported that biglycan, a small leucine-rich dermatan sulfate proteoglycan, intensifies ALK5-Smad2/3 signaling by TGF-β1 and downregulates syndecan-4 expression in vascular endothelial cells. In the present study, we investigated the crosstalk between the expressions of syndecan-1 and other proteoglycan species (syndecan-4, perlecan, glypican-1, and biglycan) in bovine aortic endothelial cells in a culture system. These data suggested that syndecan-1 downregulated syndecan-4 expression by suppressing the endogenous FGF-2-dependent ERK1/2 pathway and FGF-2-independent p38 MAPK pathway in the cells. Moreover, this crosstalk was a one-way communication from syndecan-1 to syndecan-4, suggesting that syndecan-4 compensated for the reduced activity in the regulation of vascular endothelial cell functions caused by the decreased expression of syndecan-1 under certain conditions.
Keywords: MAPK; Syndecan-1; Syndecan-4; Vascular endothelial cells.
© 2021 The Authors.
Figures
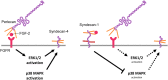

Similar articles
-
Transforming Growth Factor-β1 Modulates the Expression of Syndecan-4 in Cultured Vascular Endothelial Cells in a Biphasic Manner.J Cell Biochem. 2017 Aug;118(8):2009-2017. doi: 10.1002/jcb.25861. Epub 2017 Apr 10. J Cell Biochem. 2017. PMID: 28019669 Free PMC article.
-
Biglycan Intensifies ALK5-Smad2/3 Signaling by TGF-β1 and Downregulates Syndecan-4 in Cultured Vascular Endothelial Cells.J Cell Biochem. 2017 May;118(5):1087-1096. doi: 10.1002/jcb.25721. Epub 2017 Jan 10. J Cell Biochem. 2017. PMID: 27585241 Free PMC article.
-
Cell Density-Dependent Fibroblast Growth Factor-2 Signaling Regulates Syndecan-4 Expression in Cultured Vascular Endothelial Cells.Int J Mol Sci. 2020 May 24;21(10):3698. doi: 10.3390/ijms21103698. Int J Mol Sci. 2020. PMID: 32456321 Free PMC article.
-
Soluble vascular endothelial glycocalyx proteoglycans as potential therapeutic targets in inflammatory diseases.Immunol Cell Biol. 2024 Feb;102(2):97-116. doi: 10.1111/imcb.12712. Epub 2023 Nov 20. Immunol Cell Biol. 2024. PMID: 37982607 Review.
-
N-syndecan: structure and function of a transmembrane heparan sulfate proteoglycan.Perspect Dev Neurobiol. 1996;3(4):331-46. Perspect Dev Neurobiol. 1996. PMID: 9117264 Review.
Cited by
-
A Narrative Review: Syndecans in Aortic Aneurysm Pathogenesis and Course-Biomarkers and Targets?Int J Mol Sci. 2025 Jan 30;26(3):1211. doi: 10.3390/ijms26031211. Int J Mol Sci. 2025. PMID: 39940978 Free PMC article. Review.
-
Alterations in heparan sulfate proteoglycan synthesis and sulfation and the impact on vascular endothelial function.Matrix Biol Plus. 2022 Sep 7;16:100121. doi: 10.1016/j.mbplus.2022.100121. eCollection 2022 Dec. Matrix Biol Plus. 2022. PMID: 36160687 Free PMC article. Review.
-
Fingolimod does not prevent syndecan-4 shedding from the endothelial glycocalyx in a cultured human umbilical vein endothelial cell model of vascular injury.Intensive Care Med Exp. 2022 Aug 18;10(1):34. doi: 10.1186/s40635-022-00462-7. Intensive Care Med Exp. 2022. PMID: 35980492 Free PMC article.
-
Wooden breast myopathy is characterized by satellite cell dysfunction and syndecan-4 shedding.Front Physiol. 2024 Dec 23;15:1513311. doi: 10.3389/fphys.2024.1513311. eCollection 2024. Front Physiol. 2024. PMID: 39764382 Free PMC article.
-
No association between intravenous fluid volume and endothelial glycocalyx shedding in patients undergoing resuscitation for sepsis in the emergency department.Sci Rep. 2022 May 24;12(1):8733. doi: 10.1038/s41598-022-12752-x. Sci Rep. 2022. PMID: 35610344 Free PMC article.
References
-
- Campbell J.H., Tachas G., Black M.J., Cockerill G., Campbell G.R. Molecular biology of vascular hypertrophy. Basic Res. Cardiol. 1991;86(Suppl 1):3–11. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

