Comparison of two methods for prolong storage of decellularized mouse whole testis for tissue engineering application: An experimental study
- PMID: 33997591
- PMCID: PMC8106816
- DOI: 10.18502/ijrm.v19i4.9058
Comparison of two methods for prolong storage of decellularized mouse whole testis for tissue engineering application: An experimental study
Abstract
Background: Biological scaffolds are derived by the decellularization of tissues or organs. Various biological scaffolds, such as scaffolds for the liver, lung, esophagus, dermis, and human testicles, have been produced. Their application in tissue engineering has created the need for cryopreservation processes to store these scaffolds.
Objective: The aim was to compare the two methods for prolong storage testicular scaffolds.
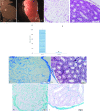
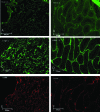
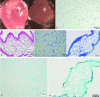
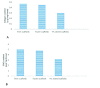
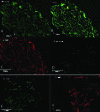
Materials and methods: In this experimental study, 20 male NMRI mice (8 wk) were sacrificed and their testes were removed and treated with 0.5% sodium dodecyl sulfate followed by Triton X-100 0.5%. The efficiency of decellularization was determined by histology and DNA quantification. Testicular scaffolds were stored in phosphate-buffered saline solution at 4°C or cryopreserved by programmed slow freezing followed by storage in liquid nitrogen. Masson's trichrome staining, Alcian blue staining and immunohistochemistry, collagen assay, and glycosaminoglycan assay were done prior to and after six months of storage under each condition.
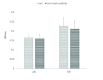
Results: Hematoxylin-eosin staining showed no remnant cells after the completion of decellularization. DNA content analysis indicated that approximately 98% of the DNA was removed from the tissue (p = 0.02). Histological evaluation confirmed the preservation of extracellular matrix components in the fresh and frozen-thawed scaffolds. Extracellular matrix components were decreased by 4°C-stored scaffolds. Cytotoxicity tests with mouse embryonic fibroblast showed that the scaffolds were biocompatible and did not have a harmful effect on the proliferation of mouse embryonic fibroblast cells.
Conclusion: Our results demonstrated the superiority of the slow freezing method for prolong storage of testicular scaffolds.
Keywords: Mouse.; Scaffold; Testis; Cryopreservation.
Copyright © 2021 Majidi Gharenaz et al.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Three-Dimensional Culture of Mouse Spermatogonial Stem Cells Using A Decellularised Testicular Scaffold.Cell J. 2020 Jan;21(4):410-418. doi: 10.22074/cellj.2020.6304. Epub 2019 Jul 29. Cell J. 2020. PMID: 31376322 Free PMC article.
-
Decellularization of the mouse ovary: comparison of different scaffold generation protocols for future ovarian bioengineering.J Ovarian Res. 2019 Jun 22;12(1):58. doi: 10.1186/s13048-019-0531-3. J Ovarian Res. 2019. PMID: 31228949 Free PMC article.
-
The Decellularized Calf Testis: Introducing Suitable Scaffolds for Spermatogenesis Studies.Int J Fertil Steril. 2023 Nov 7;18(1):32-39. doi: 10.22074/ijfs.2023.1989173.1433. Int J Fertil Steril. 2023. PMID: 38041457 Free PMC article.
-
Decellularization of kidney tissue: comparison of sodium lauryl ether sulfate and sodium dodecyl sulfate for allotransplantation in rat.Cell Tissue Res. 2021 Nov;386(2):365-378. doi: 10.1007/s00441-021-03517-5. Epub 2021 Aug 23. Cell Tissue Res. 2021. PMID: 34424397
-
Derivation and characterization of a cytocompatible scaffold from human testis.Hum Reprod. 2015 Feb;30(2):256-67. doi: 10.1093/humrep/deu330. Epub 2014 Dec 11. Hum Reprod. 2015. PMID: 25505010
Cited by
-
Influence of Storage Conditions on Decellularized Porcine Conjunctiva.Bioengineering (Basel). 2023 Mar 11;10(3):350. doi: 10.3390/bioengineering10030350. Bioengineering (Basel). 2023. PMID: 36978741 Free PMC article.
-
Decellularized Colorectal Cancer Matrices as Bioactive Scaffolds for Studying Tumor-Stroma Interactions.Cancers (Basel). 2022 Jan 12;14(2):359. doi: 10.3390/cancers14020359. Cancers (Basel). 2022. PMID: 35053521 Free PMC article. Review.
-
Decellularization of xenografted tumors provides cell-specific in vitro 3D environment.Front Oncol. 2022 Aug 18;12:956940. doi: 10.3389/fonc.2022.956940. eCollection 2022. Front Oncol. 2022. PMID: 36059712 Free PMC article.
-
Biomaterials for Testicular Bioengineering: How far have we come and where do we have to go?Front Endocrinol (Lausanne). 2023 Mar 16;14:1085872. doi: 10.3389/fendo.2023.1085872. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37008920 Free PMC article. Review.
-
Retinoic acid-releasing scaffold based on chitosan hydrogel and testis decellular plates.Bioimpacts. 2024 May 13;15:30007. doi: 10.34172/bi.30007. eCollection 2025. Bioimpacts. 2024. PMID: 39963568 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
