A new CcpA binding site plays a bidirectional role in carbon catabolism in Bacillus licheniformis
- PMID: 33997685
- PMCID: PMC8091064
- DOI: 10.1016/j.isci.2021.102400
A new CcpA binding site plays a bidirectional role in carbon catabolism in Bacillus licheniformis
Abstract
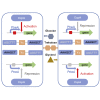
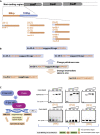
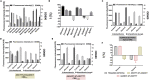
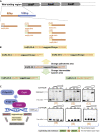
Bacillus licheniformis is widely used to produce various valuable products, such as food enzymes, industrial chemicals, and biocides. The carbon catabolite regulation process in the utilization of raw materials is crucial to maximizing the efficiency of this microbial cell factory. The current understanding of the molecular mechanism of this regulation is based on limited motif patterns in protein-DNA recognition, where the typical catabolite-responsive element (CRE) motif is "TGWNANCGNTNWCA". Here, CRETre is identified and characterized as a new CRE. It consists of two palindrome arms of 6 nucleotides (AGCTTT/AAAGCT) and an intermediate spacer. CRETre is involved in bidirectional regulation in a glucose stress environment. When AGCTTT appears in the 5' end, the regulatory element exhibits a carbon catabolite activation effect, while AAAGCT in the 5' end corresponds to carbon catabolite repression. Further investigation indicated a wide occurrence of CRETre in the genome of B. licheniformis.
Keywords: Microbial Metabolism; Microbiology; Structural Biology.
© 2021 The Author(s).
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures




References
-
- Asai K., Baik S.H., Kasahara Y., Moriya S., Ogasawara N. Regulation of the transport system for C4-dicarboxylic acids in Bacillus subtilis. Microbiology. 2000;146:263–271. - PubMed
-
- Bruckner R., Titgemeyer F. Carbon catabolite repression in bacteria: choice of the carbon source and autoregulatory limitation of sugar utilization. FEMS Microbiol. Lett. 2002;209:141–148. - PubMed
-
- Eastmond P.J., Graham I.A. Trehalose metabolism: a regulatory role for trehalose-6-phosphate? Curr. Opin. Plant Biol. 2003;6:231–235. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

