Regulation of intercellular biomolecule transfer-driven tumor angiogenesis and responses to anticancer therapies
- PMID: 33998600
- PMCID: PMC8121529
- DOI: 10.1172/JCI144225
Regulation of intercellular biomolecule transfer-driven tumor angiogenesis and responses to anticancer therapies
Abstract
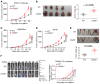
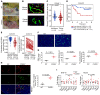
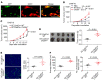
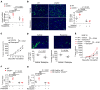
Intercellular biomolecule transfer (ICBT) between malignant and benign cells is a major driver of tumor growth, resistance to anticancer therapies, and therapy-triggered metastatic disease. Here we characterized cholesterol 25-hydroxylase (CH25H) as a key genetic suppressor of ICBT between malignant and endothelial cells (ECs) and of ICBT-driven angiopoietin-2-dependent activation of ECs, stimulation of intratumoral angiogenesis, and tumor growth. Human CH25H was downregulated in the ECs from patients with colorectal cancer and the low levels of stromal CH25H were associated with a poor disease outcome. Knockout of endothelial CH25H stimulated angiogenesis and tumor growth in mice. Pharmacologic inhibition of ICBT by reserpine compensated for CH25H loss, elicited angiostatic effects (alone or combined with sunitinib), augmented the therapeutic effect of radio-/chemotherapy, and prevented metastatic disease induced by these regimens. We propose inhibiting ICBT to improve the overall efficacy of anticancer therapies and limit their prometastatic side effects.
Keywords: Colorectal cancer; Endothelial cells; Oncology; Vascular Biology.
Conflict of interest statement
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

