Dystrophin deficiency impairs vascular structure and function in the canine model of Duchenne muscular dystrophy
- PMID: 33999411
- PMCID: PMC8713812
- DOI: 10.1002/path.5704
Dystrophin deficiency impairs vascular structure and function in the canine model of Duchenne muscular dystrophy
Abstract
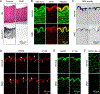
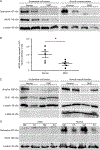
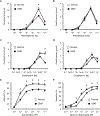
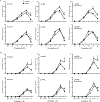
Duchenne muscular dystrophy (DMD) is a muscle-wasting disease caused by dystrophin deficiency. Vascular dysfunction has been suggested as an underlying pathogenic mechanism in DMD. However, this has not been thoroughly studied in a large animal model. Here we investigated structural and functional changes in the vascular smooth muscle and endothelium of the canine DMD model. The expression of dystrophin and endothelial nitric oxide synthase (eNOS), neuronal NOS (nNOS), and the structure and function of the femoral artery from 15 normal and 16 affected adult dogs were evaluated. Full-length dystrophin was detected in the endothelium and smooth muscle in normal but not affected dog arteries. Normal arteries lacked nNOS but expressed eNOS in the endothelium. NOS activity and eNOS expression were reduced in the endothelium of dystrophic dogs. Dystrophin deficiency resulted in structural remodeling of the artery. In affected dogs, the maximum tension induced by vasoconstrictor phenylephrine and endothelin-1 was significantly reduced. In addition, acetylcholine-mediated vasorelaxation was significantly impaired, whereas exogenous nitric oxide-induced vasorelaxation was significantly enhanced. Our results suggest that dystrophin plays a crucial role in maintaining the structure and function of vascular endothelium and smooth muscle in large mammals. Vascular defects may contribute to DMD pathogenesis. © 2021 The Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
Keywords: Duchenne muscular dystrophy; canine model; dystrophin; eNOS; nNOS; vasculature; vasoconstriction; vasorelaxation.
© 2021 The Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
Figures

Similar articles
-
Nitric oxide-dependent attenuation of noradrenaline-induced vasoconstriction is impaired in the canine model of Duchenne muscular dystrophy.J Physiol. 2018 Nov;596(21):5199-5216. doi: 10.1113/JP275672. Epub 2018 Sep 20. J Physiol. 2018. PMID: 30152022 Free PMC article.
-
Vascular endothelial dysfunction in Duchenne muscular dystrophy is restored by bradykinin through upregulation of eNOS and nNOS.Basic Res Cardiol. 2012 Jan;107(1):240. doi: 10.1007/s00395-011-0240-6. Epub 2011 Dec 23. Basic Res Cardiol. 2012. PMID: 22193759 Free PMC article.
-
Smooth muscle-specific dystrophin expression improves aberrant vasoregulation in mdx mice.Hum Mol Genet. 2006 Jul 15;15(14):2266-75. doi: 10.1093/hmg/ddl151. Epub 2006 Jun 15. Hum Mol Genet. 2006. PMID: 16777842
-
Canine models of Duchenne muscular dystrophy and their use in therapeutic strategies.Mamm Genome. 2012 Feb;23(1-2):85-108. doi: 10.1007/s00335-011-9382-y. Epub 2012 Jan 5. Mamm Genome. 2012. PMID: 22218699 Free PMC article. Review.
-
Animal models of Duchenne muscular dystrophy: from basic mechanisms to gene therapy.Dis Model Mech. 2015 Mar;8(3):195-213. doi: 10.1242/dmm.018424. Dis Model Mech. 2015. PMID: 25740330 Free PMC article. Review.
Cited by
-
Investigation of Genes and Proteins Expression Associating Serotonin Signaling Pathway in Lung and Pulmonary Artery Tissues of Dogs with Pulmonary Hypertension Secondary to Degenerative Mitral Valve Disease: The Preliminary Study.Vet Sci. 2022 Sep 28;9(10):530. doi: 10.3390/vetsci9100530. Vet Sci. 2022. PMID: 36288144 Free PMC article.
-
The importance of dystrophin and the dystrophin associated proteins in vascular smooth muscle.Front Physiol. 2022 Nov 25;13:1059021. doi: 10.3389/fphys.2022.1059021. eCollection 2022. Front Physiol. 2022. PMID: 36505053 Free PMC article. Review.
-
Re-examination of therapeutic management of muscular dystrophies using a vascular smooth muscle-centered approach.J Smooth Muscle Res. 2023;59:67-80. doi: 10.1540/jsmr.59.67. J Smooth Muscle Res. 2023. PMID: 37673649 Free PMC article.
-
Poor bone health in Duchenne muscular dystrophy: a multifactorial problem beyond corticosteroids and loss of ambulation.Front Endocrinol (Lausanne). 2024 Nov 28;15:1398050. doi: 10.3389/fendo.2024.1398050. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39669499 Free PMC article. Review.
-
Becker muscular dystrophy mice showed site-specific decay of type IIa fibers with capillary change in skeletal muscle.Elife. 2025 Mar 17;13:RP100665. doi: 10.7554/eLife.100665. Elife. 2025. PMID: 40094282 Free PMC article.
References
-
- Koenig M, Hoffman EP, Bertelson CJ, et al. Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell 1987; 50: 509–517. - PubMed
-
- Hoffman EP, Brown RH, Jr., Kunkel LM. Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 1987; 51: 919–928. - PubMed
-
- Fenichel GM. On the pathogenesis of duchenne muscular dystrophy. Dev Med Child Neurol 1975; 17: 527–533. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

