T-Cell Immunoglobulin and Mucin Domain-Containing Protein-4 Is Critical for Kupffer Cell Homeostatic Function in the Activation and Resolution of Liver Ischemia Reperfusion Injury
- PMID: 33999437
- PMCID: PMC9060306
- DOI: 10.1002/hep.31906
T-Cell Immunoglobulin and Mucin Domain-Containing Protein-4 Is Critical for Kupffer Cell Homeostatic Function in the Activation and Resolution of Liver Ischemia Reperfusion Injury
Abstract
Background and aims: Liver ischemia reperfusion injury (IRI) remains an unresolved clinical problem. This study dissected roles of liver-resident macrophage Kupffer cells (KCs), with a functional focus on efferocytosis receptor T-cell immunoglobulin and mucin domain-containing protein-4 (TIM-4), in both the activation and resolution of IRI in a murine liver partial warm ischemia model.
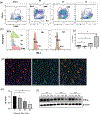
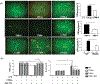
Approach and results: Fluorescence-activated cell sorting results showed that TIM-4 was expressed exclusively by KCs, but not infiltrating macrophages (iMФs), in IR livers. Anti-TIM-4 antibody depleted TIM-4+ macrophages in vivo, resulting in either alleviation or deterioration of liver IRI, which was determined by the repopulation kinetics of the KC niche with CD11b+ macrophages. To determine the KC-specific function of TIM-4, we reconstituted clodronate-liposome-treated mice with exogenous wild-type or TIM-4-deficient KCs at either 0 hour or 24 hours postreperfusion. TIM-4 deficiency in KCs resulted in not only increases in the severity of liver IRI (at 6 hours postreperfusion), but also impairment of the inflammation resolution (at 7 days postreperfusion). In vitro analysis revealed that TIM-4 promoted KC efferocytosis to regulate their Toll-like receptor response by up-regulating IL-10 and down-regulating TNF-α productions.
Conclusions: TIM-4 is critical for KC homeostatic function in both the activation and resolution of liver IRI by efferocytosis.
© 2021 by the American Association for the Study of Liver Diseases.
Conflict of interest statement
Figures




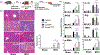

References
-
- Zhai Y, Shen Xd, O’Connell R, Gao F, Lassman C, Busuttil RW, Cheng G, et al. Cutting Edge: TLR4 Activation Mediates Liver Ischemia/Reperfusion Inflammatory Response via IFN Regulatory Factor 3-Dependent MyD88-Independent Pathway. The Journal of Immunology 2004;173:7115–7119. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

