Importance of circadian timing for aging and longevity
- PMID: 34001884
- PMCID: PMC8129076
- DOI: 10.1038/s41467-021-22922-6
Importance of circadian timing for aging and longevity
Abstract
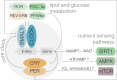
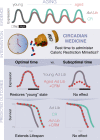
Dietary restriction (DR) decreases body weight, improves health, and extends lifespan. DR can be achieved by controlling how much and/or when food is provided, as well as by adjusting nutritional composition. Because these factors are often combined during DR, it is unclear which are necessary for beneficial effects. Several drugs have been utilized that target nutrient-sensing gene pathways, many of which change expression throughout the day, suggesting that the timing of drug administration is critical. Here, we discuss how dietary and pharmacological interventions promote a healthy lifespan by influencing energy intake and circadian rhythms.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

