Active Site Hydrogen Bonding Induced in Cytochrome P450cam by Effector Putidaredoxin
- PMID: 34006086
- PMCID: PMC8750573
- DOI: 10.1021/acs.biochem.1c00075
Active Site Hydrogen Bonding Induced in Cytochrome P450cam by Effector Putidaredoxin
Abstract
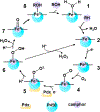
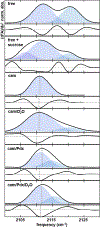
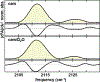
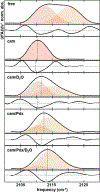
Cytochrome P450s are diverse and powerful catalysts that can activate molecular oxygen to oxidize a wide variety of substrates. Catalysis relies on effective uptake of two electrons and two protons. For cytochrome P450cam, an archetypal member of the superfamily, the second electron must be supplied by the redox partner putidaredoxin (Pdx). Pdx also plays an effector role beyond electron transfer, but after decades the mechanism remains under investigation. We applied infrared spectroscopy to heme-ligated CN- to examine the influence of Pdx binding. The results indicate that Pdx induces the population of a conformation wherein the CN- ligand forms a strong hydrogen bond to a solvent water molecule, experimentally corroborating the formation of a proposed proton delivery network. Further, characterization of T252A P450cam implicates the side chain of Thr252 in regulating the population equilibrium of hydrogen-bonded states within the P450cam/Pdx complex, which could underlie its role in directing activated oxygen toward product formation and preventing reaction uncoupling through peroxide release.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
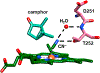
References
-
- Denisov IG, Makris TM, Sligar SG, and Schlichting I (2005) Structure and Chemistry of Cytochrome P450. Chem. Rev. 105, 2253–2277. - PubMed
-
- Shaik S, Cohen S, Wang Y, Chen H, Kumar D, and Thiel W (2010) P450 Enzymes: Their Structure, Reactivity, and Selectivity Modeled by QM/MM Calculations. Chem. Rev. 110, 949–1017. - PubMed
-
- Ortiz de Montellano PR, Ed. (2015) Cytochrome P450: Structure, Mechanism, and Biochemistry, 4th ed., Springer International Publishing, New York.
-
- Tyson CA, Lipscomb JD, and Gunsalus IC (1972) The Roles of Putidaredoxin and P450cam in Methylene Hydroxylation. J. Biol. Chem. 247, 5777–5784. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

