Kokumi taste perception is functional in a model carnivore, the domestic cat (Felis catus)
- PMID: 34006911
- PMCID: PMC8131363
- DOI: 10.1038/s41598-021-89558-w
Kokumi taste perception is functional in a model carnivore, the domestic cat (Felis catus)
Abstract
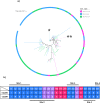
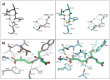
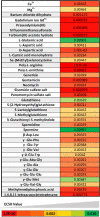
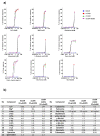
Kokumi taste is a well-accepted and characterised taste modality and is described as a sensation of enhancement of sweet, salty, and umami tastes. The Calcium Sensing Receptor (CaSR) has been designated as the putative kokumi taste receptor for humans, and a number of kokumi-active ligands of CaSR have been discovered recently with activity confirmed both in vivo and in vitro. Domestic cats (Felis catus) are obligate carnivores and accordingly, their diet is abundant in proteins, peptides, and amino acids. We hypothesised that CaSR is a key taste receptor for carnivores, due to its role in the detection of different peptides and amino acids in other species. Using in silico, in vitro and in vivo approaches, here we compare human CaSR to that of a model carnivore, the domestic cat. We found broad similarities in ligand specificity, but differences in taste sensitivity between the two species. Indeed our in vivo data shows that cats are sensitive to CaCl2 as a kokumi compound, but don't show this same activity with Glutathione, whereas for humans the reverse is true. Collectively, our data suggest that kokumi is an important taste modality for carnivores that drives the palatability of meat-derived compounds such as amino acids and peptides, and that there are differences in the perception of kokumi taste between carnivores and omnivores.
Conflict of interest statement
A.L., M.G., C.HdA., J.A., Z.N.L., D.W.L., and S.J.M. are all employees of Mars Petcare UK. All of the work presented was funded by Mars Petcare UK.
Figures


Similar articles
-
Umami taste perception and preferences of the domestic cat (Felis catus), an obligate carnivore.Chem Senses. 2023 Jan 1;48:bjad026. doi: 10.1093/chemse/bjad026. Chem Senses. 2023. PMID: 37551788 Free PMC article.
-
Kokumi substances, enhancers of basic tastes, induce responses in calcium-sensing receptor expressing taste cells.PLoS One. 2012;7(4):e34489. doi: 10.1371/journal.pone.0034489. Epub 2012 Apr 12. PLoS One. 2012. PMID: 22511946 Free PMC article.
-
Involvement of the calcium-sensing receptor in human taste perception.J Biol Chem. 2010 Jan 8;285(2):1016-22. doi: 10.1074/jbc.M109.029165. Epub 2009 Nov 5. J Biol Chem. 2010. PMID: 19892707 Free PMC article.
-
Current progress in kokumi-active peptides, evaluation and preparation methods: a review.Crit Rev Food Sci Nutr. 2022;62(5):1230-1241. doi: 10.1080/10408398.2020.1837726. Epub 2020 Oct 26. Crit Rev Food Sci Nutr. 2022. PMID: 33103468 Review.
-
Emerging roles of the extracellular calcium-sensing receptor in nutrient sensing: control of taste modulation and intestinal hormone secretion.Br J Nutr. 2014 Jun;111 Suppl 1:S16-22. doi: 10.1017/S0007114513002250. Br J Nutr. 2014. PMID: 24382107 Review.
Cited by
-
Activation of the Calcium-Sensing Receptor by a Subfraction of Amino Acids Contained in Thyroid Drainage Fluid.ACS Pharmacol Transl Sci. 2024 Jun 28;7(7):1937-1950. doi: 10.1021/acsptsci.3c00350. eCollection 2024 Jul 12. ACS Pharmacol Transl Sci. 2024. PMID: 39022353 Free PMC article.
-
The flavor-enhancing action of glutamate and its mechanism involving the notion of kokumi.NPJ Sci Food. 2023 Jan 27;7(1):3. doi: 10.1038/s41538-023-00178-2. NPJ Sci Food. 2023. PMID: 36707516 Free PMC article. Review.
-
Interkingdom Detection of Bacterial Quorum-Sensing Molecules by Mammalian Taste Receptors.Microorganisms. 2023 May 16;11(5):1295. doi: 10.3390/microorganisms11051295. Microorganisms. 2023. PMID: 37317269 Free PMC article. Review.
-
Effect of kokumi taste-active γ-glutamyl peptides on amiloride-sensitive epithelial Na+ channels in rat fungiform taste cells.Biochem Biophys Rep. 2022 Dec 1;33:101400. doi: 10.1016/j.bbrep.2022.101400. eCollection 2023 Mar. Biochem Biophys Rep. 2022. PMID: 36471792 Free PMC article.
-
Cryo-EM structure of the calcium-sensing receptor complexed with the kokumi substance γ-glutamyl-valyl-glycine.Sci Rep. 2025 Jan 31;15(1):3894. doi: 10.1038/s41598-025-87999-1. Sci Rep. 2025. PMID: 39890873 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

