Improved Performance of ELISA and Immunochromatographic Tests Using a New Chimeric A2-Based Protein for Human Visceral Leishmaniasis Diagnosis
- PMID: 34007852
- PMCID: PMC8102099
- DOI: 10.1155/2021/5568077
Improved Performance of ELISA and Immunochromatographic Tests Using a New Chimeric A2-Based Protein for Human Visceral Leishmaniasis Diagnosis
Abstract
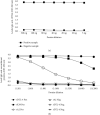
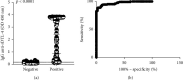
Methods: A total of 1028 sera samples were used for the development and validation of ELISA (321 samples from L. infantum-infected patients, 62 samples from VL/AIDS coinfected patients, 236 samples from patients infected with other diseases, and 409 samples from healthy donors). A total of 520 sera samples were used to develop and validate ICT (249 samples from L. infantum-infected patients, 46 samples from VL/AIDS coinfected patients, 40 samples from patients infected with other diseases, and 185 samples from healthy donors). Findings. Using the validation sera panels, DTL-4-based ELISA displayed an overall sensitivity of 94.61% (95% CI: 89.94-97.28), a specificity of 99.41% (95% CI: 96.39-99.99), and an accuracy of 97.02% (95% CI: 94.61-98.38), while for ICT, sensitivity, specificity, and accuracy values corresponded to 91.98% (95% CI: 86.65-95.39), 100.00% (95% CI: 96.30-100.00), and 95.14% (95% CI: 91.62-97.15), respectively. When testing sera samples from VL/AIDS coinfected patients, DTL-4-ELISA displayed a sensitivity of 77.42% (95% CI: 65.48-86.16), a specificity of 99.41% (95% CI: 96.39-99.99), and an accuracy of 93.51% (95% CI: 89.49%-96.10%), while for DTL-4-ICT, sensitivity was 73.91% (95% CI: 59.74-84.40), specificity was 90.63% (95% CI: 81.02-95.63), and accuracy was 82.00% (95% CI: 73.63-90.91).
Conclusion: DTL-4 is a promising candidate antigen for serodiagnosis of VL patients, including those with VL/AIDS coinfection, when incorporated into ELISA or ICT test formats.
Copyright © 2021 Maria Marta Figueiredo et al.
Conflict of interest statement
We declare that this work has not been influenced by any financial, personal, or professional interest.
Figures




Similar articles
-
Evaluation of three recombinant proteins for the development of ELISA and immunochromatographic tests for visceral leishmaniasis serodiagnosis.Mem Inst Oswaldo Cruz. 2019 Feb 4;114:e180405. doi: 10.1590/0074-02760180405. Mem Inst Oswaldo Cruz. 2019. PMID: 30726344 Free PMC article.
-
Urine- and serum-based ELISA using a new recombinant chimeric protein to diagnose tegumentary and visceral leishmaniasis: a preliminary study.Exp Parasitol. 2025 Aug;275:108980. doi: 10.1016/j.exppara.2025.108980. Epub 2025 Jun 23. Exp Parasitol. 2025. PMID: 40562158
-
Performance of rK39-based immunochromatographic rapid diagnostic test for serodiagnosis of visceral leishmaniasis using whole blood, serum and oral fluid.PLoS One. 2020 Apr 2;15(4):e0230610. doi: 10.1371/journal.pone.0230610. eCollection 2020. PLoS One. 2020. PMID: 32240188 Free PMC article.
-
A new Leishmania-specific hypothetical protein and its non-described specific B cell conformational epitope applied in the serodiagnosis of canine visceral leishmaniasis.Parasitol Res. 2016 Apr;115(4):1649-58. doi: 10.1007/s00436-016-4904-x. Epub 2016 Jan 19. Parasitol Res. 2016. PMID: 26782811 Review.
-
Potential antigenic targets used in immunological tests for diagnosis of tegumentary leishmaniasis: A systematic review.PLoS One. 2021 May 27;16(5):e0251956. doi: 10.1371/journal.pone.0251956. eCollection 2021. PLoS One. 2021. PMID: 34043671 Free PMC article.
Cited by
-
Recombinant protein KR95 as an alternative for serological diagnosis of human visceral leishmaniasis in the Americas.PLoS One. 2023 Mar 2;18(3):e0282483. doi: 10.1371/journal.pone.0282483. eCollection 2023. PLoS One. 2023. PMID: 36862710 Free PMC article.
-
Exploring Bioinformatics Solutions for Improved Leishmaniasis Diagnostic Tools: A Review.Molecules. 2024 Nov 7;29(22):5259. doi: 10.3390/molecules29225259. Molecules. 2024. PMID: 39598648 Free PMC article. Review.
-
Performance of Extracellular Vesicles From Leishmania (Leishmania) infantum for Serological Diagnosis of Human and Canine Visceral Leishmaniasis.J Parasitol Res. 2025 Jan 21;2025:8355886. doi: 10.1155/japr/8355886. eCollection 2025. J Parasitol Res. 2025. PMID: 39877665 Free PMC article.
-
ChimLeish, a new recombinant chimeric protein evaluated as a diagnostic and prognostic marker for visceral leishmaniasis and human immunodeficiency virus coinfection.Parasitol Res. 2021 Dec;120(12):4037-4047. doi: 10.1007/s00436-021-07342-1. Epub 2021 Oct 19. Parasitol Res. 2021. PMID: 34664113 Free PMC article.
-
Improved diagnostic sensitivity of human strongyloidiasis using point-of-care mixed recombinant antigen-based immunochromatography.Parasite. 2023;30:60. doi: 10.1051/parasite/2023063. Epub 2023 Dec 13. Parasite. 2023. PMID: 38099622 Free PMC article.
References
-
- Brasil, Ministério da Saúde. Leishmaniose visceral: o que é, causas, sintomas, tratamento, diagnóstico e prevenção. 2020, http://saude.gov.br/saude-de-a-z/leishmaniose-visceral.
-
- WHO. Leishmaniasis. World Health Organization; 2020. https://www.who.int/leishmaniasis/visceral_leishmaniasis/en/
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

