Postcontractile blood oxygenation level-dependent (BOLD) response in Duchenne muscular dystrophy
- PMID: 34013753
- PMCID: PMC8325615
- DOI: 10.1152/japplphysiol.00634.2020
Postcontractile blood oxygenation level-dependent (BOLD) response in Duchenne muscular dystrophy
Abstract
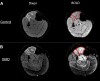
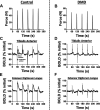
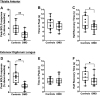
Duchenne muscular dystrophy (DMD) is characterized by a progressive replacement of muscle by fat and fibrous tissue, muscle weakness, and loss of functional abilities. Impaired vasodilatory and blood flow responses to muscle activation have also been observed in DMD and associated with mislocalization of neuronal nitric oxide synthase mu (nNOSμ) from the sarcolemma. The objective of this study was to determine whether the postcontractile blood oxygen level-dependent (BOLD) MRI response is impaired in DMD and correlated with established markers of disease severity in DMD, including MRI muscle fat fraction (FF) and clinical functional measures. Young boys with DMD (n = 16, 5-14 yr) and unaffected controls (n = 16, 5-14 yr) were evaluated using postcontractile BOLD, FF, and functional assessments. The BOLD response was measured following five brief (2 s) maximal voluntary dorsiflexion contractions, each separated by 1 min of rest. FFs from the anterior compartment lower leg muscles were quantified via chemical shift-encoded imaging. Functional abilities were assessed using the 10 m walk/run and the 6-min walk distance (6MWD). The peak BOLD responses in the tibialis anterior and extensor digitorum longus were reduced (P < 0.001) in DMD compared with controls. Furthermore, the anterior compartment peak BOLD response correlated with function (6MWD ρ = 0.87, P < 0.0001; 10 m walk/run time ρ = -0.78, P < 0.001) and FF (ρ = -0.52, P = 0.05). The reduced postcontractile BOLD response in DMD may reflect impaired microvascular function. The relationship observed between the postcontractile peak BOLD response and functional measures and FF suggests that the BOLD response is altered with disease severity in DMD.NEW & NOTEWORTHY This study examined the postcontractile blood oxygen level-dependent (BOLD) response in boys with Duchenne muscular dystrophy (DMD) and unaffected controls, and correlated this measure to markers of disease severity. Our findings indicate that the postcontractile BOLD response is impaired in DMD after brief muscle contractions, is correlated to disease severity, and may be valuable to implement in future studies to evaluate treatments targeting microvascular function in DMD.
Keywords: Duchenne muscular dystrophy; blood oxygenation level dependent; magnetic resonance imaging; microvascular; skeletal muscle.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures



References
-
- Bushby K, Finkel R, Birnkrant DJ, Case LE, Clemens PR, Cripe L, Kaul A, Kinnett K, McDonald C, Pandya S, Poysky J, Shapiro F, Tomezsko J, Constantin C; DMD Care Considerations Working Group. Diagnosis and management of Duchenne muscular dystrophy, part 1: diagnosis, and pharmacological and psychosocial management. Lancet Neurol 9: 77–93, 2010. doi: 10.1016/S1474-4422(09)70271-6. - DOI - PubMed
-
- Lai Y, Thomas GD, Yue Y, Yang HT, Li D, Long C, Judge L, Bostick B, Chamberlain JS, Terjung RL, Duan D. Dystrophins carrying spectrin-like repeats 16 and 17 anchor nNOS to the sarcolemma and enhance exercise performance in a mouse model of muscular dystrophy. J Clin Invest 119: 624–635, 2009. doi: 10.1172/JCI36612. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

