Twenty-Year Public Health Impact of 7- and 13-Valent Pneumococcal Conjugate Vaccines in US Children
- PMID: 34013855
- PMCID: PMC8153862
- DOI: 10.3201/eid2706.204238
Twenty-Year Public Health Impact of 7- and 13-Valent Pneumococcal Conjugate Vaccines in US Children
Abstract
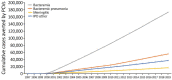
Pneumococcal conjugate vaccines (PCVs) have been used in the United States since 2000. To assess the cumulative 20-year effect of PCVs on invasive pneumococcal disease (IPD) incidence among children <5 years of age, we analyzed Active Bacterial Core Surveillance data, conducted a literature review, and modeled expected and observed disease. We found that PCVs have averted >282,000 cases of IPD, including ≈16,000 meningitis, ≈172,000 bacteremia, and ≈55,000 bacteremic pneumonia cases. In addition, vaccination has prevented 97 million healthcare visits for otitis media, 438,914-706,345 hospitalizations for pneumonia, and 2,780 total deaths. IPD cases declined 91%, from 15,707 in 1997 to 1,382 in 2019. Average annual visits for otitis media declined 41%, from 78 visits/100 children before PCV introduction to 46 visits/100 children after PCV13 introduction. Annual pneumonia hospitalizations declined 66%-79%, from 110,000-175,000 in 1997 to 37,000 in 2019. These findings confirm the substantial benefits of PCVs for preventing IPD in children.
Keywords: IPD; PCV13; PCV7; PCVs; Streptococcus pneumoniae; United States; bacteria; bacterial infections; children; incidence; invasive pneumococcal disease; literature review; otitis media; pneumococcal conjugate vaccines; pneumococcal disease; pneumococcal pneumonia; pneumococcal vaccination; vaccination; vaccine-preventable diseases; vaccines.
Figures

References
-
- Advisory Committee on Immunization Practices. Preventing pneumococcal disease among infants and young children. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2000;49(RR-9):1–35. - PubMed
-
- Centers for Disease Control and Prevention (CDC). Direct and indirect effects of routine vaccination of children with 7-valent pneumococcal conjugate vaccine on incidence of invasive pneumococcal disease—United States, 1998-2003. MMWR Morb Mortal Wkly Rep. 2005;54:893–7. - PubMed
-
- Black S, Shinefield H, Fireman B, Lewis E, Ray P, Hansen JR, et al. ; Northern California Kaiser Permanente Vaccine Study Center Group. Efficacy, safety and immunogenicity of heptavalent pneumococcal conjugate vaccine in children. Pediatr Infect Dis J. 2000;19:187–95. 10.1097/00006454-200003000-00003 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

