IL-6 Inhibition Reduces Neuronal Injury in a Murine Model of Ventilator-induced Lung Injury
- PMID: 34014798
- PMCID: PMC8525200
- DOI: 10.1165/rcmb.2021-0072OC
IL-6 Inhibition Reduces Neuronal Injury in a Murine Model of Ventilator-induced Lung Injury
Abstract
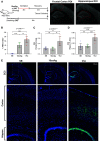
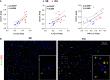
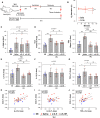
Mechanical ventilation is a known risk factor for delirium, a cognitive impairment characterized by dysfunction of the frontal cortex and hippocampus. Although IL-6 is upregulated in mechanical ventilation-induced lung injury (VILI) and may contribute to delirium, it is not known whether the inhibition of systemic IL-6 mitigates delirium-relevant neuropathology. To histologically define neuropathological effects of IL-6 inhibition in an experimental VILI model, VILI was simulated in anesthetized adult mice using a 35 cc/kg tidal volume mechanical ventilation model. There were two control groups, as follow: 1) spontaneously breathing or 2) anesthetized and mechanically ventilated with 10 cc/kg tidal volume to distinguish effects of anesthesia from VILI. Two hours before inducing VILI, mice were treated with either anti-IL-6 antibody, anti-IL-6 receptor antibody, or saline. Neuronal injury, stress, and inflammation were assessed using immunohistochemistry. CC3 (cleaved caspase-3), a neuronal apoptosis marker, was significantly increased in the frontal (P < 0.001) and hippocampal (P < 0.0001) brain regions and accompanied by significant increases in c-Fos and heat shock protein-90 in the frontal cortices of VILI mice compared with control mice (P < 0.001). These findings were not related to cerebral hypoxia, and there was no evidence of irreversible neuronal death. Frontal and hippocampal neuronal CC3 were significantly reduced with anti-IL-6 antibody (P < 0.01 and P < 0.0001, respectively) and anti-IL-6 receptor antibody (P < 0.05 and P < 0.0001, respectively) compared with saline VILI mice. In summary, VILI induces potentially reversible neuronal injury and inflammation in the frontal cortex and hippocampus, which is mitigated with systemic IL-6 inhibition. These data suggest a potentially novel neuroprotective role of systemic IL-6 inhibition that justifies further investigation.
Keywords: COVID-19; IL-6; VILI; delirium; neuronal injury.
Figures

Comment in
-
The Lung-Brain Axis in Ventilator-induced Brain Injury: Enter IL-6.Am J Respir Cell Mol Biol. 2021 Oct;65(4):339-340. doi: 10.1165/rcmb.2021-0233ED. Am J Respir Cell Mol Biol. 2021. PMID: 34153209 Free PMC article. No abstract available.
Similar articles
-
Systemic interleukin-6 inhibition ameliorates acute neuropsychiatric phenotypes in a murine model of acute lung injury.Crit Care. 2022 Sep 13;26(1):274. doi: 10.1186/s13054-022-04159-x. Crit Care. 2022. PMID: 36100846 Free PMC article.
-
Prone positioning reduces frontal and hippocampal neuronal dysfunction in a murine model of ventilator-induced lung injury.Front Med (Lausanne). 2022 Nov 4;9:987202. doi: 10.3389/fmed.2022.987202. eCollection 2022. Front Med (Lausanne). 2022. PMID: 36405620 Free PMC article.
-
Microglia ameliorate delirium-like phenotypes in a murine model of acute ventilator-induced lung injury.J Neuroinflammation. 2024 Oct 21;21(1):270. doi: 10.1186/s12974-024-03260-y. J Neuroinflammation. 2024. PMID: 39434161 Free PMC article.
-
Mechanical ventilation induces lung and brain injury through ATP production, P2Y1 receptor activation and dopamine release.Bioengineered. 2022 Feb;13(2):2346-2359. doi: 10.1080/21655979.2021.2022269. Bioengineered. 2022. PMID: 35034579 Free PMC article. Review.
-
Future directions in ventilator-induced lung injury associated cognitive impairment: a new sight.Front Physiol. 2023 Dec 18;14:1308252. doi: 10.3389/fphys.2023.1308252. eCollection 2023. Front Physiol. 2023. PMID: 38164198 Free PMC article. Review.
Cited by
-
Acute kidney injury-associated delirium: a review of clinical and pathophysiological mechanisms.Crit Care. 2022 Aug 27;26(1):258. doi: 10.1186/s13054-022-04131-9. Crit Care. 2022. PMID: 36030220 Free PMC article. Review.
-
Acute hypoxaemic respiratory failure after treatment with lower tidal volume ventilation facilitated by extracorporeal carbon dioxide removal: long-term outcomes from the REST randomised trial.Thorax. 2023 Aug;78(8):767-774. doi: 10.1136/thorax-2022-218874. Epub 2022 Oct 5. Thorax. 2023. PMID: 36198573 Free PMC article. Clinical Trial.
-
Multidimensional integrated lung protection measures attenuate pulmonary inflammation and improve lung function by inhibiting the NF-κB signaling pathway in elderly patients with lung dysfunction.Clin Exp Med. 2025 May 22;25(1):171. doi: 10.1007/s10238-025-01703-1. Clin Exp Med. 2025. PMID: 40405004 Free PMC article.
-
Could SARS-CoV-2 Spike Protein Be Responsible for Long-COVID Syndrome?Mol Neurobiol. 2022 Mar;59(3):1850-1861. doi: 10.1007/s12035-021-02696-0. Epub 2022 Jan 13. Mol Neurobiol. 2022. PMID: 35028901 Free PMC article. Review.
-
Exogenous L-fucose attenuates depression induced by chronic unpredictable stress: Implicating core fucosylation has an antidepressant potential.J Biol Chem. 2025 Mar;301(3):108230. doi: 10.1016/j.jbc.2025.108230. Epub 2025 Jan 27. J Biol Chem. 2025. PMID: 39864626 Free PMC article.
References
-
- Slutsky AS, Tremblay LN. Multiple system organ failure. Is mechanical ventilation a contributing factor? Am J Respir Crit Care Med. 1998;157:1721–1725. - PubMed
-
- Wilcox ME, Brummel NE, Archer K, Ely EW, Jackson JC, Hopkins RO. Cognitive dysfunction in ICU patients: risk factors, predictors, and rehabilitation interventions. Crit Care Med. 2013;41(9)(Suppl 1):S81–S98. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

