COVID-19 Among Patients With Multiple Sclerosis: A Systematic Review
- PMID: 34016734
- PMCID: PMC8142838
- DOI: 10.1212/NXI.0000000000001001
COVID-19 Among Patients With Multiple Sclerosis: A Systematic Review
Erratum in
-
COVID-19 Among Patients With Multiple Sclerosis: A Systematic Review.Neurol Neuroimmunol Neuroinflamm. 2021 Jul 2;8(5):e1050. doi: 10.1212/NXI.0000000000001050. Print 2021 Jul. Neurol Neuroimmunol Neuroinflamm. 2021. PMID: 34215683 Free PMC article. No abstract available.
Abstract
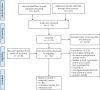
Objective: We systematically reviewed the literature on COVID-19 in patients with multiple sclerosis (MS).
Methods: We searched PubMed, Scopus, EMBASE, CINAHL, Web of Science, Google Scholar, and World Health Organization database from December 1, 2019, to December 18, 2020. Three conference abstract databases were also searched. We included any types of studies that reported characteristics of patients with MS with COVID-19.
Results: From an initial 2,679 publications and 3,138 conference abstracts, 87 studies (67 published articles and 20 abstracts) consisting of 4,310 patients with suspected/confirmed COVID-19 with MS met the inclusion criteria. The female/male ratio was 2.53:1, the mean (SD) age was 44.91 (4.31) years, the mean disease duration was 12.46 (2.27), the mean Expanded Disability Status Scale score was 2.54 (0.81), the relapsing/progressive ratio was 4.75:1, and 32.9% of patients had at least 1 comorbidity. The most common symptoms were fever (68.8%), followed by cough (63.9%), fatigue/asthenia (51.2%), and shortness of breath (39.5%). In total, 837 of 4,043 patients with MS with suspected/confirmed COVID-19 (20.7%) required hospitalization, and 130 of 4,310 (3.0%) died of COVID-19. Among suspected/confirmed patients, the highest hospitalization and mortality rates were in patients with no disease-modifying therapies (42.9% and 8.4%), followed by B cell-depleting agents (29.2% and 2.5%).
Conclusion: Our study suggested that MS did not significantly increase the mortality rate from COVID-19. These data should be interpreted with caution as patients with MS are more likely female and younger compared with the general population where age and male sex seem to be risk factors for worse disease outcome.
Copyright © 2021 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the American Academy of Neurology.
References
-
- Wijnands JM, Kingwell E, Zhu F, et al. . Infection-related health care utilization among people with and without multiple sclerosis. Mult Scler J 2017;23:1506–1516. - PubMed
-
- Montgomery S, Hillert J, Bahmanyar S. Hospital admission due to infections in multiple sclerosis patients. Eur J Neurol 2013;20:1153–1160. - PubMed
-
- OWorld Health Organization. Coronavirus Disease (COVID-19) Outbreak. Accessed September 18, 2020. who.int
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous