A Comparison of Breathing Stimulants for Reversal of Synthetic Opioid-Induced Respiratory Depression in Conscious Rats
- PMID: 34021024
- PMCID: PMC8686717
- DOI: 10.1124/jpet.121.000675
A Comparison of Breathing Stimulants for Reversal of Synthetic Opioid-Induced Respiratory Depression in Conscious Rats
Abstract
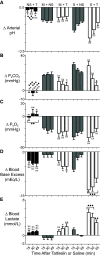
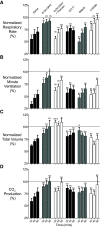
Potent synthetic opioids are an important cause of death in the United States' opioid epidemic, and a breathing stimulant may have utility in treating opioid overdose. We hypothesized that sufentanil-induced respiratory depression may be reversed by breathing stimulant administration. Using nose-only plethysmography and arterial blood analysis, we compared effects of several breathing stimulants in reversing sufentanil-induced respiratory depression in conscious rats. We studied taltirelin (1 mg/kg i.v.), PKTHPP (5 mg/kg i.v.), CX717 (30 mg/kg i.v.), BIMU8 (1 mg/kg i.v.), A85380 (30 μg/kg i.v.), and 8-hydroxy-2-(di-n-propylamino)tetralin (8-OH-DPAT) (150 μg/kg i.v./i.m.) and used sufentanil (10 μg/kg i.v.). By plethysmography (in % baseline, mean ± S.E.M.), taltirelin restored ventilation in sufentanil-treated rats (from 50 ± 5% to 102 ± 8%) by increased breathing rates (from 80 ± 4% to 160 ± 12%). By arterial blood analysis, however, taltirelin did not correct hypoxia, decreased hypercarbia only after 45 minutes, and worsened metabolic acidosis (base excess from +0 ± 1 to -7 ± 1 mEq/l). Additionally, taltirelin increased exhaled carbon dioxide, an estimate of oxygen consumption, by up to 64%. PKTHPP, CX717, BIMU8, and A85380 failed to significantly change ventilation or arterial blood values in sufentanil-treated rats. 8-OH-DPAT, however, improved ventilation (from 54 ± 8% to 92 ± 10%), reversed hypercarbia (from 64 ± 6 to 47 ± 2 mmHg), and shortened time to righting from 43 ± 4 to 15 ± 1 minutes in sufentanil-treated rats placed supine. Taltirelin has limited therapeutic potential, as its ventilatory effects are offset by metabolic acidosis, possibly from increased oxygen consumption. At the doses studied, PKTHPP, CX717, BIMU8, and A85380 have limited effects in reversing sufentanil-induced respiratory depression; 8-OH-DPAT, however, warrants further study. SIGNIFICANCE STATEMENT: Respiratory depression is an important cause of death after potent synthetic opioid overdose. 8-Hydroxy-2-(di-n-propylamino)tetralin or related compounds may be useful in treating respiratory depression as caused by potent synthetic opioids.
Copyright © 2021 by The American Society for Pharmacology and Experimental Therapeutics.
Figures





Similar articles
-
Are thyrotropin-releasing hormone (TRH) and analog taltirelin viable reversal agents of opioid-induced respiratory depression?Pharmacol Res Perspect. 2022 Jun;10(3):e00974. doi: 10.1002/prp2.974. Pharmacol Res Perspect. 2022. PMID: 35621218 Free PMC article. Review.
-
Intravenous and Intratracheal Thyrotropin Releasing Hormone and Its Analog Taltirelin Reverse Opioid-Induced Respiratory Depression in Isoflurane Anesthetized Rats.J Pharmacol Exp Ther. 2018 Jul;366(1):105-112. doi: 10.1124/jpet.118.248377. Epub 2018 Apr 19. J Pharmacol Exp Ther. 2018. PMID: 29674333 Free PMC article.
-
Serotonin therapies for opioid-induced disordered swallow and respiratory depression.J Appl Physiol (1985). 2024 Apr 1;136(4):821-843. doi: 10.1152/japplphysiol.00509.2023. Epub 2024 Feb 22. J Appl Physiol (1985). 2024. PMID: 38385184 Free PMC article.
-
The counteraction of opioid-induced ventilatory depression by the serotonin 1A-agonist 8-OH-DPAT does not antagonize antinociception in rats in situ and in vivo.Anesth Analg. 2009 Apr;108(4):1169-76. doi: 10.1213/ane.0b013e318198f828. Anesth Analg. 2009. PMID: 19299781
-
Pharmacology of viable mechanism agnostic respiratory stimulants for the reversal of drug-induced respiratory depression in humans.Expert Opin Drug Metab Toxicol. 2023 Jul-Dec;19(10):671-679. doi: 10.1080/17425255.2023.2262386. Epub 2023 Oct 27. Expert Opin Drug Metab Toxicol. 2023. PMID: 37795596 Review.
Cited by
-
New Efforts to Demonstrate the Successful Use of TRH as a Therapeutic Agent.Int J Mol Sci. 2023 Jul 4;24(13):11047. doi: 10.3390/ijms241311047. Int J Mol Sci. 2023. PMID: 37446225 Free PMC article. Review.
-
Nicotine and opioid co-dependence: Findings from bench research to clinical trials.Neurosci Biobehav Rev. 2022 Mar;134:104507. doi: 10.1016/j.neubiorev.2021.12.030. Epub 2021 Dec 27. Neurosci Biobehav Rev. 2022. PMID: 34968525 Free PMC article. Review.
-
Respiratory effects of thyrotropin-releasing hormone and its analogue taltirelin on opioid-induced respiratory depression.Br J Anaesth. 2022 Jul;129(1):e4-e6. doi: 10.1016/j.bja.2022.03.022. Epub 2022 Apr 19. Br J Anaesth. 2022. PMID: 35459532 Free PMC article. No abstract available.
-
Are thyrotropin-releasing hormone (TRH) and analog taltirelin viable reversal agents of opioid-induced respiratory depression?Pharmacol Res Perspect. 2022 Jun;10(3):e00974. doi: 10.1002/prp2.974. Pharmacol Res Perspect. 2022. PMID: 35621218 Free PMC article. Review.
References
-
- Barnett A, Goldstein J, Fiedler E, Taber R (1975) Etonitazine-induced rigidity and its antagonism by centrally acting muscle relaxants. Eur J Pharmacol 30:23–28. - PubMed
-
- Bennett JA, Abrams JT, Van Riper DF, Horrow JC (1997) Difficult or impossible ventilation after sufentanil-induced anesthesia is caused primarily by vocal cord closure. Anesthesiology 87:1070–1074. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

