Metabolomics Profiling of Patients With A-β+ Ketosis-Prone Diabetes During Diabetic Ketoacidosis
- PMID: 34021044
- PMCID: PMC8385613
- DOI: 10.2337/db21-0066
Metabolomics Profiling of Patients With A-β+ Ketosis-Prone Diabetes During Diabetic Ketoacidosis
Abstract
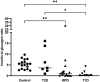
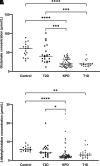
When stable and near-normoglycemic, patients with "A-β+" ketosis-prone diabetes (KPD) manifest accelerated leucine catabolism and blunted ketone oxidation, which may underlie their proclivity to develop diabetic ketoacidosis (DKA). To understand metabolic derangements in A-β+ KPD patients during DKA, we compared serum metabolomics profiles of adults during acute hyperglycemic crises, without (n = 21) or with (n = 74) DKA, and healthy control subjects (n = 17). Based on 65 kDa GAD islet autoantibody status, C-peptide, and clinical features, 53 DKA patients were categorized as having KPD and 21 type 1 diabetes (T1D); 21 nonketotic patients were categorized as having type 2 diabetes (T2D). Patients with KPD and patients with T1D had higher counterregulatory hormones and lower insulin-to-glucagon ratio than patients with T2D and control subjects. Compared with patients withT2D and control subjects, patients with KPD and patients with T1D had lower free carnitine and higher long-chain acylcarnitines and acetylcarnitine (C2) but lower palmitoylcarnitine (C16)-to-C2 ratio; a positive relationship between C16 and C2 but negative relationship between carnitine and β-hydroxybutyrate (BOHB); higher branched-chain amino acids (BCAAs) and their ketoacids but lower ketoisocaproate (KIC)-to-Leu, ketomethylvalerate (KMV)-to-Ile, ketoisovalerate (KIV)-to-Val, isovalerylcarnitine-to-KIC+KMV, propionylcarnitine-to-KIV+KMV, KIC+KMV-to-C2, and KIC-to-BOHB ratios; and lower glutamate and 3-methylhistidine. These data suggest that during DKA, patients with KPD resemble patients with T1D in having impaired BCAA catabolism and accelerated fatty acid flux to ketones-a reversal of their distinctive BCAA metabolic defect when stable. The natural history of A-β+ KPD is marked by chronic but varying dysregulation of BCAA metabolism.
© 2021 by the American Diabetes Association.
Figures
References
-
- Banerji MA, Chaiken RL, Huey H, et al. . GAD antibody negative NIDDM in adult black subjects with diabetic ketoacidosis and increased frequency of human leukocyte antigen DR3 and DR4. Flatbush diabetes. Diabetes 1994;43:741–745 - PubMed
-
- Maldonado M, Hampe CS, Gaur LK, et al. . Ketosis-prone diabetes: dissection of a heterogeneous syndrome using an immunogenetic and beta-cell functional classification, prospective analysis, and clinical outcomes. J Clin Endocrinol Metab 2003;88:5090–5098 - PubMed
-
- Mauvais-Jarvis F, Sobngwi E, Porcher R, et al. . Ketosis-prone type 2 diabetes in patients of sub-Saharan African origin: clinical pathophysiology and natural history of beta-cell dysfunction and insulin resistance. Diabetes 2004;53:645–653 - PubMed
-
- Ramos-Román MA, Piñero-Piloña A, Adams-Huet B, Raskin P. Comparison of type 1, type 2, and atypical ketosis-prone diabetes at 4 years of diabetes duration. J Diabetes Complications 2006;20:137–144 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

