Glucose and NAADP trigger elementary intracellular β-cell Ca2+ signals
- PMID: 34021189
- PMCID: PMC8140081
- DOI: 10.1038/s41598-021-88906-0
Glucose and NAADP trigger elementary intracellular β-cell Ca2+ signals
Abstract
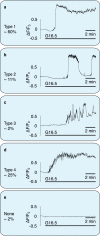
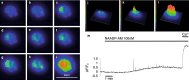
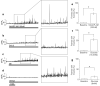
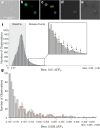
Pancreatic β-cells release insulin upon a rise in blood glucose. The precise mechanisms of stimulus-secretion coupling, and its failure in Diabetes Mellitus Type 2, remain to be elucidated. The consensus model, as well as a class of currently prescribed anti-diabetic drugs, are based around the observation that glucose-evoked ATP production in β-cells leads to closure of cell membrane ATP-gated potassium (KATP) channels, plasma membrane depolarisation, Ca2+ influx, and finally the exocytosis of insulin granules. However, it has been demonstrated by the inactivation of this pathway using genetic and pharmacological means that closure of the KATP channel alone may not be sufficient to explain all β-cell responses to glucose elevation. We have previously proposed that NAADP-evoked Ca2+ release is an important step in stimulus-secretion coupling in pancreatic β-cells. Here we show using total internal reflection fluorescence (TIRF) microscopy that glucose as well as the Ca2+ mobilising messenger nicotinic acid adenine dinucleotide phosphate (NAADP), known to operate in β-cells, lead to highly localised elementary intracellular Ca2+ signals. These were found to be obscured by measurements of global Ca2+ signals and the action of powerful SERCA-based sequestration mechanisms at the endoplasmic reticulum (ER). Building on our previous work demonstrating that NAADP-evoked Ca2+ release is an important step in stimulus-secretion coupling in pancreatic β-cells, we provide here the first demonstration of elementary Ca2+ signals in response to NAADP, whose occurrence was previously suspected. Optical quantal analysis of these events reveals a unitary event amplitude equivalent to that of known elementary Ca2+ signalling events, inositol trisphosphate (IP3) receptor mediated blips, and ryanodine receptor mediated quarks. We propose that a mechanism based on these highly localised intracellular Ca2+ signalling events mediated by NAADP may initially operate in β-cells when they respond to elevations in blood glucose.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

