Research models and mesenchymal/epithelial plasticity of osteosarcoma
- PMID: 34022967
- PMCID: PMC8141200
- DOI: 10.1186/s13578-021-00600-w
Research models and mesenchymal/epithelial plasticity of osteosarcoma
Abstract
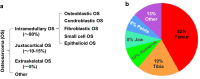
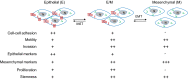
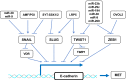
Most osteosarcomas (OSs) develop from mesenchymal cells at the bone with abnormal growth in young patients. OS has an annual incidence of 3.4 per million people and a 60-70% 5-year surviving rate. About 20% of OS patients have metastasis at diagnosis, and only 27% of patients with metastatic OS survive longer than 5 years. Mutation of tumor suppressors RB1, TP53, REQL4 and INK4a and/or deregulation of PI3K/mTOR, TGFβ, RANKL/NF-κB and IGF pathways have been linked to OS development. However, the agents targeting these pathways have yielded disappointing clinical outcomes. Surgery and chemotherapy remain the main treatments of OS. Recurrent and metastatic OSs are commonly resistant to these therapies. Spontaneous canine models, carcinogen-induced rodent models, transgenic mouse models, human patient-derived xenograft models, and cell lines from animal and human OSs have been developed for studying the initiation, growth and progression of OS and testing candidate drugs of OS. The cell plasticity regulated by epithelial-to-mesenchymal transition transcription factors (EMT-TFs) such as TWIST1, SNAIL, SLUG, ZEB1 and ZEB2 plays an important role in maintenance of the mesenchymal status and promotion of cell invasion and metastasis of OS cells. Multiple microRNAs including miR-30/9/23b/29c/194/200, proteins including SYT-SSX1/2 fusion proteins and OVOL2, and other factors that inhibit AMF/PGI and LRP5 can suppress either the expression or activity of EMT-TFs to increase epithelial features and inhibit OS metastasis. Further understanding of the molecular mechanisms that regulate OS cell plasticity should provide potential targets and therapeutic strategies for improving OS treatment.
Keywords: EMT-TFs; EMT/MET-related process; Experimental model; Metastasis; Osteosarcoma.
Conflict of interest statement
All authors do not have any conflict of interests to disclose.
Figures
Similar articles
-
MicroRNA-34a targets epithelial to mesenchymal transition-inducing transcription factors (EMT-TFs) and inhibits breast cancer cell migration and invasion.Oncotarget. 2017 Mar 28;8(13):21362-21379. doi: 10.18632/oncotarget.15214. Oncotarget. 2017. PMID: 28423483 Free PMC article.
-
Downregulation of microRNA-145 promotes epithelial-mesenchymal transition via regulating Snail in osteosarcoma.Cancer Gene Ther. 2017 Feb;24(2):83-88. doi: 10.1038/cgt.2017.1. Epub 2017 Feb 10. Cancer Gene Ther. 2017. PMID: 28186090
-
Ovol2 induces mesenchymal-epithelial transition via targeting ZEB1 in osteosarcoma.Onco Targets Ther. 2018 May 22;11:2963-2973. doi: 10.2147/OTT.S157119. eCollection 2018. Onco Targets Ther. 2018. PMID: 29872308 Free PMC article.
-
Melatonin and Its Role in the Epithelial-to-Mesenchymal Transition (EMT) in Cancer.Cancers (Basel). 2024 Feb 27;16(5):956. doi: 10.3390/cancers16050956. Cancers (Basel). 2024. PMID: 38473317 Free PMC article. Review.
-
The Role of Epithelial-Mesenchymal Transition in Osteosarcoma Progression: From Biology to Therapy.Diagnostics (Basel). 2025 Mar 6;15(5):644. doi: 10.3390/diagnostics15050644. Diagnostics (Basel). 2025. PMID: 40075892 Free PMC article. Review.
Cited by
-
Decoding the Impact of Tumor Microenvironment in Osteosarcoma Progression and Metastasis.Cancers (Basel). 2023 Oct 23;15(20):5108. doi: 10.3390/cancers15205108. Cancers (Basel). 2023. PMID: 37894474 Free PMC article. Review.
-
TGF‑β inhibitor RepSox suppresses osteosarcoma via the JNK/Smad3 signaling pathway.Int J Oncol. 2021 Nov;59(5):84. doi: 10.3892/ijo.2021.5264. Epub 2021 Sep 17. Int J Oncol. 2021. PMID: 34533199 Free PMC article.
-
Establishment of orthotopic osteosarcoma animal models in immunocompetent rats through muti-rounds of in-vivo selection.BMC Cancer. 2024 Jun 7;24(1):703. doi: 10.1186/s12885-024-12361-z. BMC Cancer. 2024. PMID: 38849717 Free PMC article.
-
RNA 5-methylcytosine status is associated with DNMT2/TRDMT1 nuclear localization in osteosarcoma cell lines.J Bone Oncol. 2022 Jul 30;36:100448. doi: 10.1016/j.jbo.2022.100448. eCollection 2022 Oct. J Bone Oncol. 2022. PMID: 35942470 Free PMC article.
-
Oncogenic Long Noncoding RNAs in Prostate Cancer, Osteosarcoma, and Metastasis.Biomedicines. 2023 Feb 20;11(2):633. doi: 10.3390/biomedicines11020633. Biomedicines. 2023. PMID: 36831169 Free PMC article. Review.
References
-
- Cokkinides V, Albano J, Samuels A, Ward M, Thum J. American cancer society: cancer facts and figures. Atlanta: American Cancer Society; 2005.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

