Cryopreservation and Preparation of Thawed Spermatozoa from Rhesus Macaques (Macaca mulatta) for In Vitro Fertilization
- PMID: 34024310
- PMCID: PMC9390610
- DOI: 10.30802/AALAS-JAALAS-20-000028
Cryopreservation and Preparation of Thawed Spermatozoa from Rhesus Macaques (Macaca mulatta) for In Vitro Fertilization
Abstract
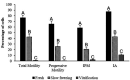
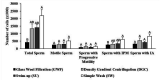
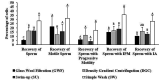
Advances in assisted reproductive technologies in rhesus macaques have allowed the development of valuable models of human disease, particularly when combined with recent techniques for gene editing. While the ability to perform in vitro fertilization (IVF) in rhesus macaques is well established, this procedure has not yet been optimized. Specifically, damage to the sperm caused by cryopreservation (cryodamage) may lead to unsuccessful artificial insemination and low fertilization and blastocyst formation rates in vitro. To address this, we systematically assessed 2 cryopreservation methods and 4 recovery methods in the following 3 interdependent experiments: 1) comparing sperm survival after vitrification or slow-freezing; 2) comparing simple wash (SW), density gradient centrifugation (DGC), swim-up (SU), and glass wool filtration (GWF) for removal of cryoprotectants and isolation of motile sperm after thawing; and 3) evaluating the efficacy for IVF of the 2 best methods of isolating thawed sperm. We found that after vitrification, only 1.2 ± 0.3% of thawed sperm were motile, whereas after slow-freezing, 42 ± 5% of thawed sperm were motile. SW was significantly better than all other isolation methods for the recovery of total sperm and for the recovery of sperm with an intact plasma membrane. The isolation methods had no significant differences in the recovery of motile sperm or sperm with progressive motility. However, IVF of ova with sperm recovered by DGC resulted in 5% more embryos and 25% more blastocysts than did IVF with sperm recovered by SW. Although additional studies are required to optimize sperm cryopreservation in rhesus macaques, our study showed that slow-freezing, coupled with DGC, provided the highest efficacy in providing functional sperm for in vitro use.
Figures
References
-
- Aalseth EP, Saacke RG. 1986. Vital staining and acrosomal evaluation of bovine sperm. Gamete Res 15:73–81. 10.1002/mrd.1120150108. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

