Combination of Immunotherapy and Radiotherapy for Recurrent Malignant Gliomas: Results From a Prospective Study
- PMID: 34025640
- PMCID: PMC8138184
- DOI: 10.3389/fimmu.2021.632547
Combination of Immunotherapy and Radiotherapy for Recurrent Malignant Gliomas: Results From a Prospective Study
Abstract
Background: World Health Organization (WHO) grade IV glioma remains one of the most lethal tumors with a dismal prognosis and inevitable recurrence. We evaluated the safety and efficacy of immunotherapy with radiotherapy in this population of patients.
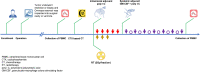
Methods: This study was a single-arm, open-label, phase I trial based on patients with recurrent WHO grade IV glioma. Patients were treated with intracranial and systemic immunoadjuvants in combination with low-dose reirradiation. The primary endpoint of the present trial was safety. Secondary endpoints were overall survival (OS) and progression-free survival (PFS). This trial is registered at ClinicalTrials.gov, NCT03392545.
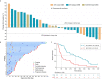
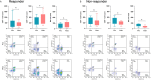
Results: Thirty patients were enrolled. The most common adverse events (AEs) were fever (66.7%), vomiting (33.3%), headache (30.0%), and fatigue (23.3%). Only a single patient experienced grade 3 fever, and no grade 4 AEs or deaths related to treatment were observed. Of the 30 patients, 1 (3.3%) had a complete response, 5 (16.7%) had a partial response, 9 (30.0%) had stable disease, and 15 (50.0%) had progressive disease, resulting in an objective response rate of 20.0%. The median PFS of the entire cohort was 88.0 (61.0-254.0) days, and the median OS was 362.0 (197.0-601.0) days. Patients could be divided into responders and non-responders, and these groups exhibited a significant difference in terms of survival time, T lymphocyte subsets, frequency of cell division cycle 27 (CDC27) mutation status, and CD15 and CD68 expression (P<0.05).
Conclusion: The combination of immunotherapy and radiotherapy is well tolerated and may provide clinical benefit for patients with recurrent WHO grade IV glioma. A prospective phase II study is needed to further validate the efficacy of our therapeutic regimen.
Keywords: immuno-oncology; immunoadjuvant; immunotherapy; malignant gliomas; reirradiation.
Copyright © 2021 Jiang, Yu, Cui, Ren, Li, Yang, Zhao, Zhu and Lin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
ERGO2: A Prospective, Randomized Trial of Calorie-Restricted Ketogenic Diet and Fasting in Addition to Reirradiation for Malignant Glioma.Int J Radiat Oncol Biol Phys. 2020 Nov 15;108(4):987-995. doi: 10.1016/j.ijrobp.2020.06.021. Epub 2020 Jun 30. Int J Radiat Oncol Biol Phys. 2020. PMID: 32619561 Clinical Trial.
-
Phase II Study of Bevacizumab and Vorinostat for Patients with Recurrent World Health Organization Grade 4 Malignant Glioma.Oncologist. 2018 Feb;23(2):157-e21. doi: 10.1634/theoncologist.2017-0501. Epub 2017 Nov 13. Oncologist. 2018. PMID: 29133513 Free PMC article. Clinical Trial.
-
Reirradiation with concurrent bevacizumab for recurrent high-grade gliomas in adult patients.Cancer Radiother. 2018 Feb;22(1):9-16. doi: 10.1016/j.canrad.2017.06.013. Epub 2017 Dec 6. Cancer Radiother. 2018. PMID: 29217134
-
Clinical efficacy of tumor antigen-pulsed DC treatment for high-grade glioma patients: evidence from a meta-analysis.PLoS One. 2014 Sep 12;9(9):e107173. doi: 10.1371/journal.pone.0107173. eCollection 2014. PLoS One. 2014. PMID: 25215607 Free PMC article.
-
Survival outcomes in pediatric recurrent high-grade glioma: results of a 20-year systematic review and meta-analysis.J Neurooncol. 2018 Mar;137(1):103-110. doi: 10.1007/s11060-017-2701-8. Epub 2017 Dec 4. J Neurooncol. 2018. PMID: 29204840 Free PMC article.
Cited by
-
Harnessing the abscopal effect for gastrointestinal malignancies in the era of immunotherapy.J Gastrointest Oncol. 2023 Jun 30;14(3):1613-1625. doi: 10.21037/jgo-23-105. Epub 2023 Jun 2. J Gastrointest Oncol. 2023. PMID: 37435204 Free PMC article. Review.
-
Advances in the field of developing biomarkers for re-irradiation: a how-to guide to small, powerful data sets and artificial intelligence.Expert Rev Precis Med Drug Dev. 2024;9(1):3-16. doi: 10.1080/23808993.2024.2325936. Epub 2024 Mar 11. Expert Rev Precis Med Drug Dev. 2024. PMID: 38550554 Free PMC article.
-
Glioma-derived ANXA1 suppresses the immune response to TLR3 ligands by promoting an anti-inflammatory tumor microenvironment.Cell Mol Immunol. 2024 Jan;21(1):47-59. doi: 10.1038/s41423-023-01110-0. Epub 2023 Dec 4. Cell Mol Immunol. 2024. PMID: 38049523 Free PMC article.
-
Safety and efficacy of radiotherapy/chemoradiotherapy combined with immune checkpoint inhibitors for non-small cell lung cancer: A systematic review and meta-analysis.Front Immunol. 2023 Mar 13;14:1065510. doi: 10.3389/fimmu.2023.1065510. eCollection 2023. Front Immunol. 2023. PMID: 36993952 Free PMC article.
-
Toll-like receptors and toll-like receptor-targeted immunotherapy against glioma.J Hematol Oncol. 2021 Oct 29;14(1):176. doi: 10.1186/s13045-021-01191-2. J Hematol Oncol. 2021. PMID: 34715891 Free PMC article. Review.
References
-
- Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, et al. . Effects of Radiotherapy With Concomitant and Adjuvant Temozolomide Versus Radiotherapy Alone on Survival in Glioblastoma in a Randomised Phase III Study: 5-Year Analysis of the EORTC-NCIC Trial. Lancet Oncol (2009) 10:459–66. 10.1016/S1470-2045(09)70025-7 - DOI - PubMed
-
- Stupp R, Taillibert S, Kanner A, Read W, Steinberg D, Lhermitte B, et al. . Effect of Tumor-Treating Fields Plus Maintenance Temozolomide vs Maintenance Temozolomide Alone on Survival in Patients With Glioblastoma: A Randomized Clinical Trial. JAMA (2017) 318:2306–16. 10.1001/jama.2017.18718 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

