Crystal structure of tris-(2-di-cyclo-hexyl-phosphino-2',6'-dimeth-oxy-1,1'-biphenyl-κ P)-μ-oxoethenyl-idene- triangulo-trigold(I) bis-(tri-fluoro-methane-sulfon-yl)imide
- PMID: 34026260
- PMCID: PMC8100268
- DOI: 10.1107/S2056989021003844
Crystal structure of tris-(2-di-cyclo-hexyl-phosphino-2',6'-dimeth-oxy-1,1'-biphenyl-κ P)-μ-oxoethenyl-idene- triangulo-trigold(I) bis-(tri-fluoro-methane-sulfon-yl)imide
Abstract
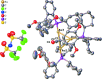
The title ketenyl-idene, [Au3(C2O)(C26H35O2P)3](C2F6NO4S2), was obtained upon exposure of [2-(di-cyclo-hexyl-phosphino)-2',6'-dimeth-oxy-1,1'-biphen-yl]gold(I) bis-(tri-fluoro-methane-sulfon-yl)imide to acetic anhydride at elevated temperature. The ketenyl-idene bridge caps the tri-gold cluster. The title compound has provided crystals that upon analysis represent the first tri-gold ketenyl-idene with atomic distances indicative of bonding inter-action between the gold atoms.
Keywords: crystal structure; ketenylidene; tri-gold.
© Hartgerink et al. 2021.
Figures

References
-
- Bogdan, P. L., Sabat, M., Sunshine, S. A., Woodcock, C. & Shriver, D. F. (1988). Inorg. Chem. 27, 1904–1910.
-
- Bondi, A. (1964). J. Phys. Chem. 68, 441–451.
-
- Bruker (2013). APEX2, SAINT and SADABS. Bruker AXS Inc. Madison, Wisconsin, USA.
-
- Ching, S., Holt, E. M., Kolis, J. W. & Shriver, D. F. (1988). Organometallics, 7, 892–898.
-
- Ching, S., Sabat, M. & Shriver, D. F. (1989). Organometallics, 8, 1047–1058.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
