Isothiourea-catalysed enantioselective Michael addition of N-heterocyclic pronucleophiles to α,β-unsaturated aryl esters
- PMID: 34040717
- PMCID: PMC8133005
- DOI: 10.1039/c9sc04303a
Isothiourea-catalysed enantioselective Michael addition of N-heterocyclic pronucleophiles to α,β-unsaturated aryl esters
Abstract
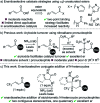
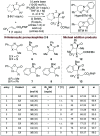
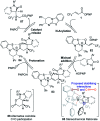
The isothiourea-catalysed enantioselective Michael addition of 3-aryloxindole and 4-substituted-dihydropyrazol-3-one pronucleophiles to α,β-unsaturated p-nitrophenyl esters is reported. This process generates products containing two contiguous stereocentres, one quaternary, in good yields and excellent enantioselectivities (>30 examples, up to > 95 : 5 dr and 99 : 1 er). This protocol harnesses the multifunctional ability of p-nitrophenoxide to promote effective catalysis. In contrast to previous methodologies using tertiary amine Lewis bases, in which the pronucleophile was used as the solvent, this work allows bespoke pronucleophiles to be used in stoichiometric quantities.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

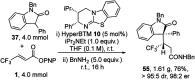
References
-
-
For selected reviews see:
- d'Angelo J. Desmaële D. Dumas F. Guingant A. Tetrahedron: Asymmetry. 1992;3:459. doi: 10.1016/S0957-4166(00)80251-7. - DOI
- Jha S. C. Joshi N. N. ARKIVOC. 2002:167.
- Bhanja C. Jena S. Nayak S. Mohapatra S. Beilstein J. Org. Chem. 2012;8:1668. - PMC - PubMed
- Zhang Y. Wang W. Catal. Sci. Technol. 2012;2:42. doi: 10.1039/C1CY00334H. - DOI
- Heravi M. M. Hajiabbasi P. Hamidi H. Curr. Org. Chem. 2014;18:489. doi: 10.2174/13852728113176660149. - DOI
- Nayak S. Chakroborty S. Bhakta S. Panda P. Mohapatra S. Res. Chem. Intermed. 2016;42:2731. doi: 10.1007/s11164-015-2193-0. - DOI
- Hui C. Pu F. Xu J. Chem.–Eur. J. 2017;23:4023. doi: 10.1002/chem.201604110. - DOI - PubMed
- Yumiko S. Mini-Rev. Org. Chem. 2018;15:236. doi: 10.2174/1570193X15666171211165344. - DOI - PMC - PubMed
-
-
-
For selected examples see:
- Xie J.-W. Yue L. Xue D. Ma X.-L. Chen Y.-C. Wu Y. Zhu J. Deng J.-G. Chem. Commun. 2006:1563. doi: 10.1039/B600647G. - DOI - PubMed
- Patil M. P. Sunoj R. B. Chem. Asian J. 2009;4:714. doi: 10.1002/asia.200800351. - DOI - PubMed
- Huang X. Wen Y.-H. Zhou F.-T. Chen C. Xu D.-C. Xie J.-W. Tetrahedron Lett. 2010;51:6637. doi: 10.1016/j.tetlet.2010.10.055. - DOI
- Mager I. Zeitler K. Org. Lett. 2010;12:1480. doi: 10.1021/ol100166z. - DOI - PubMed
- Mao Z. Jia Y. Li W. Wang R. J. Org. Chem. 2010;75:7428. doi: 10.1021/jo101188m. - DOI - PubMed
- Wang J. F. Qi C. Ge Z. M. Cheng T. M. Li R. T. Chem. Commun. 2010;46:2124. doi: 10.1039/B923925A. - DOI - PubMed
- Wang J. F. Wang X. Ge Z. M. Cheng T. M. Li R. T. Chem. Commun. 2010;46:1751. doi: 10.1039/B915852A. - DOI - PubMed
- Patora-Komisarska K. Benohoud M. Ishikawa H. Seebach D. Hayashi Y. Helv. Chim. Acta. 2011;94:719. doi: 10.1002/hlca.201100122. - DOI
- Quintard A. Alexakis A. Chem. Commun. 2011;47:7212. doi: 10.1039/C1CC11967B. - DOI - PubMed
- Vera S. Liu Y. K. Marigo M. Escudero-Adan E. C. Melchiorre P. Synlett. 2011:489.
- Lu N. Meng L. Chen D. Zhang G. J. Phys. Chem. A. 2012;116:670. doi: 10.1021/jp209308a. - DOI - PubMed
- Yoon H. S. Ho X. H. Jang J. Lee H. J. Kim S. J. Jang H. Y. Org. Lett. 2012;14:3272. doi: 10.1021/ol3011858. - DOI - PubMed
- Gu Y. Wang Y. Yu T. Y. Liang Y. M. Xu P. F. Angew. Chem., Int. Ed. 2014;53:14128. doi: 10.1002/anie.201406786. - DOI - PubMed
- Lagoutte R. Besnard C. Alexakis A. Eur. J. Org. Chem. 2016:4372. doi: 10.1002/ejoc.201600707. - DOI
- Ueda A. Umeno T. Doi M. Akagawa K. Kudo K. Tanaka M. J. Org. Chem. 2016;81:6343. doi: 10.1021/acs.joc.6b00982. - DOI - PubMed
- Jia Z. L. Wang Y. Zhao C. G. Zhang X. H. Xu P. F. Org. Lett. 2017;19:2130. doi: 10.1021/acs.orglett.7b00767. - DOI - PubMed
- Larnaud F. Sulpizi A. Haggman N. O. Hughes J. M. E. Dewez D. F. Gleason J. L. Eur. J. Org. Chem. 2017:2637. doi: 10.1002/ejoc.201700469. - DOI
- Wang Z.-X. Zhou Z. Xiao W. Ouyang Q. Du W. Chen Y.-C. Chem.–Eur. J. 2017;23:10678. doi: 10.1002/chem.201702183. - DOI - PubMed
- Mondal B. Balha M. Pan S. C. Asian J. Org. Chem. 2018;7:1788. doi: 10.1002/ajoc.201800316. - DOI
- Swiderek K. Nodling A. R. Tsai Y. H. Luk L. Y. P. Moliner V. J. Phys. Chem. A. 2018;122:451. doi: 10.1021/acs.jpca.7b11803. - DOI - PMC - PubMed
-
-
-
For an excellent overview see:
- Doyle A. G. Jacobsen E. N. Chem. Rev. 2007;107:5713. doi: 10.1021/cr068373r. - DOI - PubMed
-
. For select examples see:
- Kobayashi Y. Taniguchi Y. Hayama N. Inokuma T. Takemoto Y. Angew. Chem., Int. Ed. 2013;52:11114. doi: 10.1002/anie.201305492. - DOI - PubMed
- Inokuma T. Hoashi Y. Takemoto Y. J. Am. Chem. Soc. 2006;128:9413. doi: 10.1021/ja061364f. - DOI - PubMed
-
-
-
For selected seminal examples see:
- Kobayashi S. Suda S. Yamada M. Mukaiyama T. Chem. Lett. 1994:97. doi: 10.1246/cl.1994.97. - DOI
- Bernardi A. Karamfilova K. Sanguinetti S. Scolastico C. Tetrahedron. 1997;53:13009. doi: 10.1016/S0040-4020(97)00825-9. - DOI
- Kitajima H. Katsuki T. Synlett. 1997:568. doi: 10.1055/s-1997-3235. - DOI
- Evans D. A. Rovis T. Kozlowski M. C. Tedrow J. S. J. Am. Chem. Soc. 1999;121:1994. doi: 10.1021/ja983864h. - DOI
- Evans D. A. Willis M. C. Johnston J. N. Org. Lett. 1999;1:865. doi: 10.1021/ol9901570. - DOI - PubMed
- Evans D. A. Scheidt K. Johnston J. N. Willis M. C. J. Am. Chem. Soc. 2001;123:4480. doi: 10.1021/ja010302g. - DOI - PubMed
-
LinkOut - more resources
Full Text Sources

