Targeted Axillary Dissection in Node-Positive Breast Cancer: A Retrospective Study and Cost Analysis
- PMID: 34040910
- PMCID: PMC8139537
- DOI: 10.7759/cureus.14610
Targeted Axillary Dissection in Node-Positive Breast Cancer: A Retrospective Study and Cost Analysis
Abstract
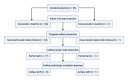
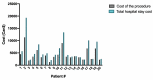
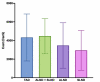
Introduction Targeted axillary dissection (TAD) is a novel technique in the field of surgical oncology. During TAD, patients with node-positive breast cancer who clinically responded to neoadjuvant chemotherapy undergo resection of a previously proven metastatic node together with sentinel lymph node dissection (SLND). We aimed to assess the success rates of seed insertion and seed retrieval in the Canadian setting, as well as hospital costs of the procedure. Methods Patients converted to clinically node-negative status post-neoadjuvant chemotherapy underwent TAD. Before surgery, an iodine-125 radioactive seed was inserted in the previously proven metastatic node. The seed node was resected together with an SLND. Axillary lymph node dissection (ALND) was performed in all patients with residual metastases. Results Radioactive seeds were successfully inserted in 34/35 patients. In 34 patients, the targeted node was successfully resected with the radioactive probe during TAD. In one patient, the seed was retrieved inferiorly in the axilla during surgery. There was no adverse event. In total, 50% (17/34) of patients had no residual metastases and were able to avoid ALND. Eight out of 17 patients who underwent ALND did not have any residual disease in their specimen. The mean cost of TAD was 25% superior to the mean cost of ALND (p = 0.02). However, the mean total cost of the hospital stay for TAD was 20% superior to the mean cost of ALND (p = 0.11). The mean cost of TAD was 4,322 Can$ (Canadian dollars), similar to the mean cost of both ALND and SLND performed during the same procedure (4,479 Can$). Conclusions TAD was successful in 97% of patients. Despite increased procedural costs, with a lesser impact on total hospital stay costs, TAD was beneficial in 50% of patients. These patients avoided the unnecessary morbidity associated with ALND.
Keywords: axillary lymph node dissection; breast cancer; breast cancer management; iodine seed; neoadjuvant chemotherapy; nodal metastases; radioactive seed; targeted axillary node dissection.
Copyright © 2021, Beniey et al.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Implementation of the Targeted Axillary Dissection Procedure in Clinically Node-Positive Breast Cancer: A Retrospective Analysis.Ann Surg Oncol. 2024 Jul;31(7):4477-4486. doi: 10.1245/s10434-024-15182-3. Epub 2024 Mar 24. Ann Surg Oncol. 2024. PMID: 38523225
-
Improved Axillary Evaluation Following Neoadjuvant Therapy for Patients With Node-Positive Breast Cancer Using Selective Evaluation of Clipped Nodes: Implementation of Targeted Axillary Dissection.J Clin Oncol. 2016 Apr 1;34(10):1072-8. doi: 10.1200/JCO.2015.64.0094. Epub 2016 Jan 25. J Clin Oncol. 2016. PMID: 26811528 Free PMC article.
-
Feasibility and accuracy of targeted axillary dissection in breast cancer patients; single center experience.Front Surg. 2024 Jan 10;10:1332142. doi: 10.3389/fsurg.2023.1332142. eCollection 2023. Front Surg. 2024. PMID: 38269392 Free PMC article.
-
Staging of women with breast cancer after introduction of sentinel node guided axillary dissection.Dan Med J. 2012 Jul;59(7):B4475. Dan Med J. 2012. PMID: 22759850 Review.
-
Improved axillary staging of breast cancer with sentinel lymphadenectomy.Ann Surg. 1995 Sep;222(3):394-9; discussion 399-401. doi: 10.1097/00000658-199509000-00016. Ann Surg. 1995. PMID: 7677468 Free PMC article. Review.
Cited by
-
Technetium-99-Guided Axillary Lymph Node Identification: A Case Report of a Novel Technique for Targeted Lymph Node Excision Biopsy for Node Positive Breast Cancer After Neoadjuvant Chemotherapy.J Med Cases. 2023 Dec;14(12):419-425. doi: 10.14740/jmc4172. Epub 2023 Dec 29. J Med Cases. 2023. PMID: 38186556 Free PMC article.
-
Assessing the Efficacy of Radioactive Iodine Seed Localisation in Targeted Axillary Dissection for Node-Positive Early Breast Cancer Patients Undergoing Neoadjuvant Systemic Therapy: A Systematic Review and Pooled Analysis.Diagnostics (Basel). 2024 Jun 2;14(11):1175. doi: 10.3390/diagnostics14111175. Diagnostics (Basel). 2024. PMID: 38893701 Free PMC article. Review.
-
Systematic review of targeted axillary dissection in node-positive breast cancer treated with neoadjuvant systemic therapy: variation in type of marker and timing of placement.Br J Surg. 2024 Mar 2;111(3):znae071. doi: 10.1093/bjs/znae071. Br J Surg. 2024. PMID: 38531689 Free PMC article.
-
Wire-Free Targeted Axillary Dissection: A Pooled Analysis of 1300+ Cases Post-Neoadjuvant Systemic Therapy in Node-Positive Early Breast Cancer.Cancers (Basel). 2024 Jun 7;16(12):2172. doi: 10.3390/cancers16122172. Cancers (Basel). 2024. PMID: 38927878 Free PMC article. Review.
-
Patient-Reported Outcomes of Omission of Breast Surgery Following Neoadjuvant Systemic Therapy: A Nonrandomized Clinical Trial.JAMA Netw Open. 2023 Sep 5;6(9):e2333933. doi: 10.1001/jamanetworkopen.2023.33933. JAMA Netw Open. 2023. PMID: 37707811 Free PMC article. Clinical Trial.
References
-
- Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC Trial. Mansel RE, Fallowfield L, Kissin M, et al. J Natl Cancer Inst. 2006;98:599–560. - PubMed
-
- A randomized comparison of sentinel-node biopsy with routine axillary dissection in breast cancer. Veronesi U, Paganelli G, Viale G, et al. N Engl J Med. 2003;349:546–553. - PubMed
-
- Surgical complications associated with sentinel lymph node dissection (SLND) plus axillary lymph node dissection compared with SLND alone in the American College of Surgeons Oncology Group Trial Z0011. Lucci A, McCall LM, Beitsch PD, et al. J Clin Oncol. 2007;25:3657–3663. - PubMed
-
- Axillary dissection versus no axillary dissection in patients with breast cancer and sentinel-node micrometastases (IBCSG 23-01): 10-year follow-up of a randomised, controlled phase 3 trial. Galimberti V, Cole BF, Viale G, et al. Lancet Oncol. 2018;19:1385–1393. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
