Regulation of stress-provoked aggressive behavior using endocannabinoids
- PMID: 34041309
- PMCID: PMC8144478
- DOI: 10.1016/j.ynstr.2021.100337
Regulation of stress-provoked aggressive behavior using endocannabinoids
Abstract
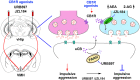
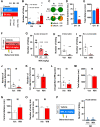
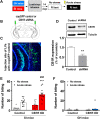
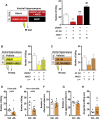
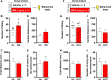
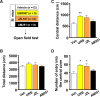
Reactive impulsive aggression is characterized by outbursts of rage and violence when subjects encounter threatening stressful events. Although impulsive aggression and violence create a high-cost burden on health and society, relatively little is known about treatment. Early adolescent social isolation (SI) alters brain development and functions. It induces hyper-excitatory in the ventral hippocampus (vHip) to promote acute stress-provoked outbursts of aggression, referred to as impulsive aggression, in mouse models. Cannabinoid type 1 receptors (CB1Rs) act on presynaptic sites and suppress neurotransmitter release into synapses. Given that CB1R activation inhibits neurotransmitter releases and modulates excitatory network activity, we tested the hypothesis that CB1R activation reduces impulsive aggression in SI mice through decreasing excitatory activity in the vHip. Here, we report that CB1R agonists, WIN-552122 (WIN) or arachidonylcyclopropylamide (ACPA), ameliorated acute stress-provoked attack behavior in the resident-intruder test without affecting general locomotion activity. Increasing endocannabinoids (eCBs) by inhibiting degradation enzymes in the vHip reduced impulsive aggression, and the effect was blunted by administration of AM251, a CB1R antagonist. Acute stress in SI mice induced c-Fos expression, a marker of neuronal activation, on vHip neurons projecting to the ventromedial hypothalamus (VMH), a well-known brain area that controls attack behavior. eCB augmentation inhibited c-Fos expression in VMH-projecting vHip neurons surrounded by CB1Rs. These results suggest that enhancing eCB signaling in order to activate CB1Rs suppresses impulsive aggression via suppressing vHip→VMH neural activity and point to a role of CB1R activation in ameliorating impulsive aggression in adults who have had adverse experiences during early adolescence.
Keywords: Acute stress; Aggression; CB1 receptor; Endocannabinoid; Hippocampus; Social isolation.
© 2021 The Authors.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
The Ventral Hippocampus Controls Stress-Provoked Impulsive Aggression through the Ventromedial Hypothalamus in Post-Weaning Social Isolation Mice.Cell Rep. 2019 Jul 30;28(5):1195-1205.e3. doi: 10.1016/j.celrep.2019.07.005. Cell Rep. 2019. PMID: 31365864
-
The dorsal raphe-to-ventral hippocampal projection modulates reactive aggression through 5-HT1B receptors.Eur J Pharmacol. 2024 Oct 15;981:176918. doi: 10.1016/j.ejphar.2024.176918. Epub 2024 Aug 17. Eur J Pharmacol. 2024. PMID: 39159717
-
Cannabidiol attenuates aggressive behavior induced by social isolation in mice: Involvement of 5-HT1A and CB1 receptors.Prog Neuropsychopharmacol Biol Psychiatry. 2019 Aug 30;94:109637. doi: 10.1016/j.pnpbp.2019.109637. Epub 2019 May 2. Prog Neuropsychopharmacol Biol Psychiatry. 2019. PMID: 31054943
-
Type-1 cannabinoid receptor signaling in neuronal development.Pharmacology. 2012;90(1-2):19-39. doi: 10.1159/000339075. Epub 2012 Jul 3. Pharmacology. 2012. PMID: 22776780 Review.
-
5-HT1A and 5-HT1B receptor agonists and aggression: a pharmacological challenge of the serotonin deficiency hypothesis.Eur J Pharmacol. 2005 Dec 5;526(1-3):125-39. doi: 10.1016/j.ejphar.2005.09.065. Epub 2005 Nov 28. Eur J Pharmacol. 2005. PMID: 16310183 Review.
Cited by
-
Chronic ingestion of soy peptide supplementation reduces aggressive behavior and abnormal fear memory caused by juvenile social isolation.Sci Rep. 2024 May 21;14(1):11557. doi: 10.1038/s41598-024-62534-w. Sci Rep. 2024. PMID: 38773352 Free PMC article.
-
URB597 induces subtle changes to aggression in adult Lister Hooded rats.Front Psychiatry. 2022 Aug 12;13:885146. doi: 10.3389/fpsyt.2022.885146. eCollection 2022. Front Psychiatry. 2022. PMID: 36032247 Free PMC article.
-
Blue light at night produces stress-evoked heightened aggression by enhancing brain-derived neurotrophic factor in the basolateral amygdala.Neurobiol Stress. 2023 Dec 15;28:100600. doi: 10.1016/j.ynstr.2023.100600. eCollection 2024 Jan. Neurobiol Stress. 2023. PMID: 38187456 Free PMC article.
-
The endocannabinoidome-gut microbiome-brain axis as a novel therapeutic target for autism spectrum disorder.J Biomed Sci. 2025 Jul 2;32(1):60. doi: 10.1186/s12929-025-01145-7. J Biomed Sci. 2025. PMID: 40605060 Free PMC article. Review.
References
-
- Bluett R.J., Baldi R., Haymer A., Gaulden A.D., Hartley N.D., Parrish W.P., Baechle J., Marcus D.J., Mardam-Bey R., Shonesy B.C., Uddin M.J., Marnett L.J., Mackie K., Colbran R.J., Winder D.G., Patel S. Endocannabinoid signalling modulates susceptibility to traumatic stress exposure. Nat. Commun. 2017;8:14782. doi: 10.1038/ncomms14782. - DOI - PMC - PubMed
-
- Centanni S.W., Morris B.D., Luchsinger J.R., Bedse G., Fetterly T.L., Patel S., Winder D.G. Endocannabinoid control of the insular-bed nucleus of the stria terminalis circuit regulates negative affective behavior associated with alcohol abstinence. Neuropsychopharmacology. 2019;44(3):526–537. doi: 10.1038/s41386-018-0257-8. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

