The squamous cell carcinoma cell line OM-1 retains both p75-dependent stratified epithelial progenitor potential and cancer stem cell properties
- PMID: 34041369
- PMCID: PMC8144356
- DOI: 10.1016/j.bbrep.2021.101003
The squamous cell carcinoma cell line OM-1 retains both p75-dependent stratified epithelial progenitor potential and cancer stem cell properties
Abstract
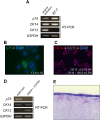
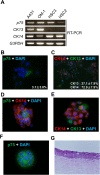
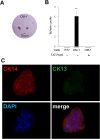
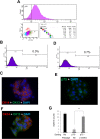
The low-affinity nerve growth factor receptor p75 is a stratified epithelial stem/progenitor marker of human epithelia. We found OM-1, a human squamous cell carcinoma (SCC) cell line, showed distinct cells with p75 cluster, especially located at the center of a growing colony in a monolayer culture. A cell with p75 cluster was surrounded by cytokeratin 14- and cytokeratin 13-expressing cells that settled at the outer margin of the colony. OM-1 cells were also capable of forming tumor spheres in a cell suspension culture, an ability which was attenuated by the inhibition of p75-signaling. Intriguingly, we also found a p75-negative cell population from a growing culture of OM-1 that re-committed to become p75-clustering cells. These results indicated the possibility that SCC with epithelial multi-layering capacity can exploit the p75-dependent stratified epithelial progenitor property for the cancer stemness.
Keywords: Cancer stem cell (CSC); Squamous cell carcinoma (SCC); p75.
© 2021 Published by Elsevier B.V.
Conflict of interest statement
The authors declare no conflicts of interest associated with this manuscript.
Figures
References
-
- Seino S., Shigeishi H., Hashikata M., Higashikawa K., Tobiume K., Uetsuki R., Ishida Y., Sasaki K., Naruse T., Rahman M.Z., Ono S., Simasue H., Ohta K., Sugiyama M., Takechi M. CD44high/ALDH1high head and neck squamous cell carcinoma cells exhibit mesenchymal characteristics and GSK3β-dependent cancer stem cell properties. J. Oral Pathol. Med. 2016;45:180–188. doi: 10.1111/jop.12348. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

