Genetic Variation in PFKFB3 Impairs Antifungal Immunometabolic Responses and Predisposes to Invasive Pulmonary Aspergillosis
- PMID: 34044589
- PMCID: PMC8263003
- DOI: 10.1128/mBio.00369-21
Genetic Variation in PFKFB3 Impairs Antifungal Immunometabolic Responses and Predisposes to Invasive Pulmonary Aspergillosis
Abstract
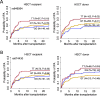
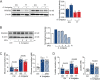
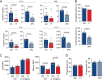
Activation of immune cells in response to fungal infection involves the reprogramming of their cellular metabolism to support antimicrobial effector functions. Although metabolic pathways such as glycolysis are known to represent critical regulatory nodes in antifungal immunity, it remains undetermined whether these are differentially regulated at the interindividual level. In this study, we identify a key role for 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 (PFKFB3) in the immunometabolic responses to Aspergillus fumigatus. A genetic association study performed in 439 recipients of allogeneic hematopoietic stem cell transplantation (HSCT) and corresponding donors revealed that the donor, but not recipient, rs646564 variant in the PFKFB3 gene increased the risk of invasive pulmonary aspergillosis (IPA) after transplantation. The risk genotype impaired the expression of PFKFB3 by human macrophages in response to fungal infection, which was correlated with a defective activation of glycolysis and the ensuing antifungal effector functions. In patients with IPA, the risk genotype was associated with lower concentrations of cytokines in the bronchoalveolar lavage fluid samples. Collectively, these findings demonstrate the important contribution of genetic variation in PFKFB3 to the risk of IPA in patients undergoing HSCT and support its inclusion in prognostic tools to predict the risk of fungal infection in this clinical setting. IMPORTANCE The fungal pathogen Aspergillus fumigatus can cause severe and life-threatening forms of infection in immunocompromised patients. Activation of glycolysis is essential for innate immune cells to mount effective antifungal responses. In this study, we report the contribution of genetic variation in the key glycolytic activator 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 (PFKFB3) to the risk of invasive pulmonary aspergillosis (IPA) after allogeneic hematopoietic stem cell transplantation. The PFKFB3 genotype associated with increased risk of infection was correlated with an impairment of the antifungal effector functions of macrophages in vitro and in patients with IPA. This work highlights the clinical relevance of genetic variation in PFKFB3 to the risk of IPA and supports its integration in risk stratification and preemptive measures for patients at high risk of IPA.
Keywords: Aspergillus; PFKFB3; antifungal immunity; immunometabolism; invasive pulmonary aspergillosis; macrophage; single nucleotide polymorphism; stem cell transplantation.
Figures
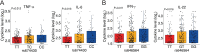
References
-
- Herbrecht R, Patterson TF, Slavin MA, Marchetti O, Maertens J, Johnson EM, Schlamm HT, Donnelly JP, Pappas PG. 2015. Application of the 2008 definitions for invasive fungal diseases to the trial comparing voriconazole versus amphotericin B for therapy of invasive aspergillosis: a collaborative study of the Mycoses Study Group (MSG 05) and the European Organization for Research and Treatment of Cancer Infectious Diseases Group. Clin Infect Dis 60:713–720. doi: 10.1093/cid/ciu911. - DOI - PubMed
-
- Maertens JA, Raad II, Marr KA, Patterson TF, Kontoyiannis DP, Cornely OA, Bow EJ, Rahav G, Neofytos D, Aoun M, Baddley JW, Giladi M, Heinz WJ, Herbrecht R, Hope W, Karthaus M, Lee DG, Lortholary O, Morrison VA, Oren I, Selleslag D, Shoham S, Thompson GR, III, Lee M, Maher RM, Schmitt-Hoffmann AH, Zeiher B, Ullmann AJ. 2016. Isavuconazole versus voriconazole for primary treatment of invasive mould disease caused by Aspergillus and other filamentous fungi (SECURE): a phase 3, randomised-controlled, non-inferiority trial. Lancet 387:760–769. doi: 10.1016/S0140-6736(15)01159-9. - DOI - PubMed
-
- van der Linden JW, Snelders E, Kampinga GA, Rijnders BJ, Mattsson E, Debets-Ossenkopp YJ, Kuijper EJ, Van Tiel FH, Melchers WJ, Verweij PE. 2011. Clinical implications of azole resistance in Aspergillus fumigatus, The Netherlands, 2007–2009. Emerg Infect Dis 17:1846–1854. doi: 10.3201/eid1710.110226. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

