Persistence with Early-Line Abatacept versus Tumor Necrosis Factor-Inhibitors for Rheumatoid Arthritis Complicated by Poor Prognostic Factors
- PMID: 34046511
- PMCID: PMC8133796
- DOI: 10.36469/jheor.2021.23684
Persistence with Early-Line Abatacept versus Tumor Necrosis Factor-Inhibitors for Rheumatoid Arthritis Complicated by Poor Prognostic Factors
Abstract
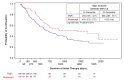
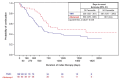
Background: Rheumatoid arthritis (RA) is a chronic inflammatory disease characterized by joint swelling and destruction that leads to severe disability. There are no clear guidelines regarding the order of therapies. Gathering data on treatment patterns outside of a clinical trial setting can provide useful context for clinicians. Objectives: To assess real-world treatment persistence in early-line abatacept versus tumor necrosis factor-inhibitors (TNFi) treated patients with RA complicated by poor prognostic factors (including anti-cyclic citrullinated peptide antibodies [ACPA] and rheumatoid factor [RF] seropositivity). Methods: We performed a multi-center retrospective medical record review. Adult patients with RA complicated by poor prognostic factors were treated with either abatacept or TNFis as the first biologic treatment at the clinic. Poor prognostic factors included ACPA+, RF+, increased C-reactive protein levels, elevated erythrocyte sedimentation rate levels, or presence of joint erosions. We report 12-month treatment persistence, time to discontinuation, reasons for discontinuation, and risk of discontinuation between patients on abatacept versus TNFi. Select results among the subgroup of ACPA+ and/or RF+ patients are presented. Results: Data on 265 patients (100 abatacept, 165 TNFis) were collected. At 12 months, 83% of abatacept patients were persistent versus 66.1% of TNFi patients (P=0.003). Median time to discontinuation was 1423 days for abatacept versus 690 days for TNFis (P=0.014). In adjusted analyses, abatacept patients had a lower risk of discontinuing index treatment due to disease progression (0.3 [95% confidence interval (CI): 0.1-0.6], P=0.001). Among the subgroup of ACPA+ and/or RF+ patients (55 abatacept, 108 TNFis), unadjusted 12-month treatment persistence was greater (83.6% versus 64.8%, P=0.012) and median time to discontinuation was longer (961 days versus 581 days, P=0.048) in abatacept versus TNFi patients. Discussion: Patients with RA complicated by poor prognostic factors taking abatacept, including the subgroup of patients with ACPA and RF seropositivity, had statistically significantly higher 12-month treatment persistence and a longer time to discontinuation than patients on TNFis. Conclusions: In a real-world setting, RA patients treated with abatacept were more likely to stay on treatment longer and had a lower risk of discontinuation than patients treated with TNFis.
Keywords: abatacept; disease modifying antirheumatic drugs; rheumatoid arthritis; therapy persistence; tumor necrosis factor inhibitors.
Figures
Similar articles
-
Association Between Baseline Anti-cyclic Citrullinated Peptide Antibodies and 6-Month Clinical Response Following Abatacept or TNF Inhibitor Treatment: A Real-World Analysis of Biologic-Experienced Patients with RA.Rheumatol Ther. 2021 Jun;8(2):937-953. doi: 10.1007/s40744-021-00310-2. Epub 2021 May 28. Rheumatol Ther. 2021. PMID: 34047953 Free PMC article.
-
Real-World Persistence and Effectiveness of Upadacitinib versus Other Janus Kinase Inhibitors and Tumor Necrosis Factor Inhibitors in Australian Patients with Rheumatoid Arthritis.Rheumatol Ther. 2025 Feb;12(1):173-202. doi: 10.1007/s40744-024-00736-4. Epub 2025 Jan 6. Rheumatol Ther. 2025. PMID: 39757285 Free PMC article.
-
Comparison of infection-related hospitalization risk and costs in tumor necrosis factor inhibitor-experienced patients with rheumatoid arthritis (RA) treated with abatacept or other targeted disease-modifying anti-rheumatic drugs (tDMARDs).J Med Econ. 2020 Sep;23(9):1025-1031. doi: 10.1080/13696998.2020.1772271. Epub 2020 Jun 13. J Med Econ. 2020. PMID: 32427547
-
Changes of anti-citrullinated peptide antibodies titers after biologic treatment in patients with rheumatoid arthritis: a systematic literature review and retrospective study.Clin Exp Rheumatol. 2023 Jul;41(7):1417-1426. doi: 10.55563/clinexprheumatol/1h6h71. Epub 2022 Dec 7. Clin Exp Rheumatol. 2023. PMID: 36533995
-
Presence of anti-cyclic citrullinated peptide antibodies is associated with better treatment response to abatacept but not to TNF inhibitors in patients with rheumatoid arthritis: a meta-analysis.Clin Exp Rheumatol. 2020 May-Jun;38(3):455-466. Epub 2019 Nov 16. Clin Exp Rheumatol. 2020. PMID: 31770089
Cited by
-
Real-world clinical outcomes and rationale for initiating abatacept as a first-line biologic for patients with anticitrullinated protein antibody- and rheumatoid factor-positive rheumatoid arthritis.J Comp Eff Res. 2024 Dec;13(12):e230144. doi: 10.57264/cer-2023-0144. Epub 2024 Nov 18. J Comp Eff Res. 2024. PMID: 39556028 Free PMC article.
References
-
- 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Aletaha Daniel, Neogi Tuhina, Silman Alan J., Funovits Julia, Felson David T., Bingham Clifton O., III, Birnbaum Neal S., Burmester Gerd R., Bykerk Vivian P., Cohen Marc D., Combe Bernard, Costenbader Karen H., Dougados Maxime, Emery Paul, Ferraccioli Gianfranco, Hazes Johanna M. W., Hobbs Kathryn, Huizinga Tom W. J., Kavanaugh Arthur, Kay Jonathan, Kvien Tore K., Laing Timothy, Mease Philip, Ménard Henri A., Moreland Larry W., Naden Raymond L., Pincus Theodore, Smolen Josef S., Stanislawska-Biernat Ewa, Symmons Deborah, Tak Paul P., Upchurch Katherine S., Vencovský Jiří, Wolfe Frederick, Hawker Gillian. Aug 10;2010 Arthritis & Rheumatism. 62(9):2569–2581. doi: 10.1002/art.27584. doi: 10.1002/art.27584. - DOI - DOI - PubMed
-
- Update on the epidemiology, risk factors, and disease outcomes of rheumatoid arthritis. van der Woude Diane, van der Helm-van Mil Annette H.M. Apr;2018 Best Practice & Research Clinical Rheumatology. 32(2):174–187. doi: 10.1016/j.berh.2018.10.005. doi: 10.1016/j.berh.2018.10.005. - DOI - DOI - PubMed
-
- Is the incidence of rheumatoid arthritis rising?: results from Olmsted County, Minnesota, 1955-2007. Myasoedova Elena, Crowson Cynthia S., Kremers Hilal Maradit, Therneau Terry M., Gabriel Sherine E. Feb 26;2010 Arthritis & Rheumatism. 62(6):1576–1582. doi: 10.1002/art.27425. doi: 10.1002/art.27425. - DOI - DOI - PMC - PubMed
-
- Clinical identification and treatment of a rapidly progressing disease state in patients with rheumatoid arthritis. Emery P., McInnes I.B., van Vollenhoven R., Kraan M.C. 2008Rheumatology (Oxford) 47(4):392–398. doi: 10.1093/rheumatology/kem257. doi: 10.1093/rheumatology/kem257. - DOI - DOI - PubMed
-
- Progression of joint damage in early rheumatoid arthritis: association with HLA–DRB1, rheumatoid factor, and anti–citrullinated protein antibodies in relation to different treatment strategies. de Vries-Bouwstra J. K., Goekoop-Ruiterman Y. P. M., Verpoort K. N., Schreuder G. M. T., Ewals J. A. P. M., Terwiel J. P., Ronday H. K., Kerstens P. J. S. M., Toes R. E. M., de Vries R. R. P., Breedveld F. C., Dijkmans B. A. C., Huizinga T. W. J., Allaart C. F. 2008Arthritis & Rheumatism. 58(5):1293–1298. doi: 10.1002/art.23439. doi: 10.1002/art.23439. - DOI - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
