Functional Analysis of Deoxyhexose Sugar Utilization in Escherichia coli Reveals Fermentative Metabolism under Aerobic Conditions
- PMID: 34047632
- PMCID: PMC8315171
- DOI: 10.1128/AEM.00719-21
Functional Analysis of Deoxyhexose Sugar Utilization in Escherichia coli Reveals Fermentative Metabolism under Aerobic Conditions
Abstract
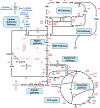
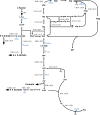
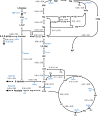
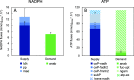
l-Rhamnose and l-fucose are the two main 6-deoxyhexoses Escherichia coli can use as carbon and energy sources. Deoxyhexose metabolism leads to the formation of lactaldehyde, whose fate depends on oxygen availability. Under anaerobic conditions, lactaldehyde is reduced to 1,2-propanediol, whereas under aerobic conditions, it should be oxidized into lactate and then channeled into the central metabolism. However, although this all-or-nothing view is accepted in the literature, it seems overly simplistic since propanediol is also reported to be present in the culture medium during aerobic growth on l-fucose. To clarify the functioning of 6-deoxyhexose sugar metabolism, a quantitative metabolic analysis was performed to determine extra- and intracellular fluxes in E. coli K-12 MG1655 (a laboratory strain) and in E. coli Nissle 1917 (a human commensal strain) during anaerobic and aerobic growth on l-rhamnose and l-fucose. As expected, lactaldehyde is fully reduced to 1,2-propanediol under anoxic conditions, allowing complete reoxidation of the NADH produced by glyceraldehyde-3-phosphate-dehydrogenase. We also found that net ATP synthesis is ensured by acetate production. More surprisingly, lactaldehyde is also primarily reduced into 1,2-propanediol under aerobic conditions. For growth on l-fucose, 13C-metabolic flux analysis revealed a large excess of available energy, highlighting the need to better characterize ATP utilization processes. The probiotic E. coli Nissle 1917 strain exhibits similar metabolic traits, indicating that they are not the result of the K-12 strain's prolonged laboratory use. IMPORTANCE E. coli's ability to survive in, grow in, and colonize the gastrointestinal tract stems from its use of partially digested food and hydrolyzed glycosylated proteins (mucins) from the intestinal mucus layer as substrates. These include l-fucose and l-rhamnose, two 6-deoxyhexose sugars, whose catabolic pathways have been established by genetic and biochemical studies. However, the functioning of these pathways has only partially been elucidated. Our quantitative metabolic analysis provides a comprehensive picture of 6-deoxyhexose sugar metabolism in E. coli under anaerobic and aerobic conditions. We found that 1,2-propanediol is a major by-product under both conditions, revealing the key role of fermentative pathways in 6-deoxyhexose sugar metabolism. This metabolic trait is shared by both E. coli strains studied here, a laboratory strain and a probiotic strain. Our findings add to our understanding of E. coli's metabolism and of its functioning in the bacterium's natural environment.
Keywords: anaerobic catabolic pathways; carbon metabolism; metabolism.
Figures

Similar articles
-
Metabolism of L-fucose and L-rhamnose in Escherichia coli: aerobic-anaerobic regulation of L-lactaldehyde dissimilation.J Bacteriol. 1988 Jan;170(1):416-21. doi: 10.1128/jb.170.1.416-421.1988. J Bacteriol. 1988. PMID: 3275622 Free PMC article.
-
Evolution of L-1, 2-propanediol catabolism in Escherichia coli by recruitment of enzymes for L-fucose and L-lactate metabolism.J Bacteriol. 1974 Apr;118(1):83-8. doi: 10.1128/jb.118.1.83-88.1974. J Bacteriol. 1974. PMID: 4595205 Free PMC article.
-
Disruption of the fucose pathway as a consequence of genetic adaptation to propanediol as a carbon source in Escherichia coli.J Bacteriol. 1976 Jun;126(3):1166-72. doi: 10.1128/jb.126.3.1166-1172.1976. J Bacteriol. 1976. PMID: 181364 Free PMC article.
-
Towards a systems level understanding of the oxygen response of Escherichia coli.Adv Microb Physiol. 2014;64:65-114. doi: 10.1016/B978-0-12-800143-1.00002-6. Adv Microb Physiol. 2014. PMID: 24797925 Review.
-
The metabolism of 6-deoxyhexoses in bacterial and animal cells.Biochimie. 1998 Nov;80(11):923-31. doi: 10.1016/s0300-9084(00)88889-6. Biochimie. 1998. PMID: 9893952 Review.
Cited by
-
A comprehensive review on microbial production of 1,2-propanediol: micro-organisms, metabolic pathways, and metabolic engineering.Biotechnol Biofuels. 2021 Nov 18;14(1):216. doi: 10.1186/s13068-021-02067-w. Biotechnol Biofuels. 2021. PMID: 34794503 Free PMC article. Review.
-
Investigation on L-rhamnose metabolism of Loigolactobacillus coryniformis subsp. coryniformis DSM 20001 and its propionate-containing fermentates.Appl Environ Microbiol. 2025 Jan 31;91(1):e0161324. doi: 10.1128/aem.01613-24. Epub 2024 Dec 18. Appl Environ Microbiol. 2025. PMID: 39692501 Free PMC article.
-
L-Rhamnose Globally Changes the Transcriptome of Planktonic and Biofilm Escherichia coli Cells and Modulates Biofilm Growth.Microorganisms. 2024 Sep 19;12(9):1911. doi: 10.3390/microorganisms12091911. Microorganisms. 2024. PMID: 39338585 Free PMC article.
-
L-Fucose is involved in human-gut microbiome interactions.Appl Microbiol Biotechnol. 2023 Jun;107(12):3869-3875. doi: 10.1007/s00253-023-12527-y. Epub 2023 May 6. Appl Microbiol Biotechnol. 2023. PMID: 37148338 Review.
-
Developing a Fluorescent Inducible System for Free Fucose Quantification in Escherichia coli.Biosensors (Basel). 2023 Mar 15;13(3):388. doi: 10.3390/bios13030388. Biosensors (Basel). 2023. PMID: 36979599 Free PMC article.
References
-
- Cheng CC, Duar RM, Lin X, Perez-Munoz ME, Tollenaar S, Oh J-H, van Pijkeren J-P, Li F, van Sinderen D, Gänzle MG, Walter J. 2020. Ecological importance of cross-feeding of the intermediate metabolite 1,2-propanediol between bacterial gut symbionts. Appl Environ Microbiol 86:e00190-20. 10.1128/AEM.00190-20. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

