Bronchial angiogenesis in horses with severe asthma and its response to corticosteroids
- PMID: 34048095
- PMCID: PMC8295704
- DOI: 10.1111/jvim.16159
Bronchial angiogenesis in horses with severe asthma and its response to corticosteroids
Abstract
Background: Severe asthma in horses is characterized by structural changes that thicken the lower airway wall, a change that is only partially reversible by current treatments. Increased vascularization contributes to the thickening of the bronchial wall in humans with asthma and is considered a potential new therapeutic target.
Objective: To determine the presence of angiogenesis in the bronchi of severely asthmatic horses, and if present, to evaluate its reversibility by treatment with corticosteroids.
Animals: Study 1: Bronchial samples from asthmatic horses in exacerbation (7), in remission (7), and aged-matched healthy horses. Study 2: Endobronchial biopsy samples from asthmatic horses in exacerbation (6) and healthy horses (6) before and after treatment with dexamethasone.
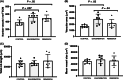
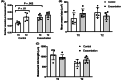
Methods: Blinded, randomized controlled study. Immunohistochemistry was performed using collagen IV as a marker for vascular basement membranes. Number of vessels, vascular area, and mean vessel size in the bronchial lamina propria were measured by histomorphometry. Reversibility of vascular changes in Study 2 was assessed after 2 weeks of treatment with dexamethasone.
Results: The number of vessels and vascular area were increased in the airway walls of asthmatic horses in exacerbation (P = .01 and P = .02, respectively) and in remission (P = .02 and P = .04, respectively) when compared to controls. In Study 2, the differences observed between groups disappeared after 2 weeks of treatment with corticosteroids because of the increased number of vessels in healthy horses.
Conclusions and clinical importance: Angiogenesis contributes to thickening of the airway wall in asthmatic horses and was not reversed by a 2-week treatment with corticosteroids.
Keywords: angiogenesis; asthma; collagen IV; horses; immunohistochemistry.
© 2021 The Authors. Journal of Veterinary Internal Medicine published by Wiley Periodicals LLC on behalf of American College of Veterinary Internal Medicine.
Conflict of interest statement
Authors declare no conflict of interest.
Figures



Similar articles
-
Bronchial vessel density is correlated with airway smooth muscle cell proliferation in horses with mild and moderate asthma.Am J Vet Res. 2025 Jan 9;86(3):ajvr.24.10.0318. doi: 10.2460/ajvr.24.10.0318. Print 2025 Mar 1. Am J Vet Res. 2025. PMID: 39787707
-
Nebulisation of dexamethasone sodium phosphate for the treatment of severe asthmatic horses.Equine Vet J. 2019 Sep;51(5):641-645. doi: 10.1111/evj.13091. Epub 2019 Mar 28. Equine Vet J. 2019. PMID: 30849189
-
Vascularity in asthmatic airways: relation to inhaled steroid dose.Thorax. 1999 Apr;54(4):289-95. doi: 10.1136/thx.54.4.289. Thorax. 1999. PMID: 10092688 Free PMC article.
-
Therapeutic approach to vascular remodelling in asthma.Pulm Pharmacol Ther. 2007;20(1):1-8. doi: 10.1016/j.pupt.2005.10.009. Epub 2005 Dec 9. Pulm Pharmacol Ther. 2007. PMID: 16338153 Review.
-
Glucocorticoid treatment in horses with asthma: A narrative review.J Vet Intern Med. 2021 Jul;35(4):2045-2057. doi: 10.1111/jvim.16189. Epub 2021 Jun 3. J Vet Intern Med. 2021. PMID: 34085342 Free PMC article. Review.
Cited by
-
Severe asthma in horses is associated with increased airway innervation.J Vet Intern Med. 2024 Jan-Feb;38(1):485-494. doi: 10.1111/jvim.16941. Epub 2023 Dec 6. J Vet Intern Med. 2024. PMID: 38054207 Free PMC article.
-
Co-Occurrence of Severe Equine Asthma and Palatal Disorders in Privately Owned Pleasure Horses.Animals (Basel). 2023 Jun 12;13(12):1962. doi: 10.3390/ani13121962. Animals (Basel). 2023. PMID: 37370472 Free PMC article.
References
-
- Pirie RS, Couetil LL, Robinson NE, et al. Equine asthma: an appropriate, translational and comprehendible terminology? Equine Vet J. 2016;48:403‐405. - PubMed
-
- Pirie RS. Recurrent airway obstruction: a review. Equine Vet J. 2014;46:276‐288. - PubMed
-
- Bullone M, Lavoie J‐P. Asthma “of horses and men”—how can equine heaves help us better understand human asthma immunopathology and its functional consequences? Mol Immunol. 2015;66:97‐105. - PubMed
-
- Bullone M, Lavoie J‐P. The equine asthma model of airway remodeling: from a veterinary to a human perspective. Cell Tissue Res. 2020;380:223‐236. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

