Rationally designed bacterial consortia to treat chronic immune-mediated colitis and restore intestinal homeostasis
- PMID: 34050144
- PMCID: PMC8163890
- DOI: 10.1038/s41467-021-23460-x
Rationally designed bacterial consortia to treat chronic immune-mediated colitis and restore intestinal homeostasis
Abstract
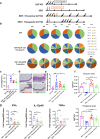
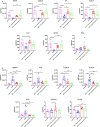
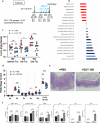
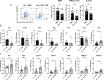
Environmental factors, mucosal permeability and defective immunoregulation drive overactive immunity to a subset of resident intestinal bacteria that mediate multiple inflammatory conditions. GUT-103 and GUT-108, live biotherapeutic products rationally designed to complement missing or underrepresented functions in the dysbiotic microbiome of IBD patients, address upstream targets, rather than targeting a single cytokine to block downstream inflammation responses. GUT-103, composed of 17 strains that synergistically provide protective and sustained engraftment in the IBD inflammatory environment, prevented and treated chronic immune-mediated colitis. Therapeutic application of GUT-108 reversed established colitis in a humanized chronic T cell-mediated mouse model. It decreased pathobionts while expanding resident protective bacteria; produced metabolites promoting mucosal healing and immunoregulatory responses; decreased inflammatory cytokines and Th-1 and Th-17 cells; and induced interleukin-10-producing colonic regulatory cells, and IL-10-independent homeostatic pathways. We propose GUT-108 for treating and preventing relapse for IBD and other inflammatory conditions characterized by unbalanced microbiota and mucosal permeability.
Conflict of interest statement
D.v.d.L., S.T., and C.H. are co-founders and shareholders with a personal financial interest in Gusto Global LLC. The results described in this manuscript support PCT patent application WO 2018/187272 Al and provisional patent application US63,094,655. R.B.S. received research grant support from Gusto Global LLC and serves as an unpaid consultant to the company. The other authors do not have competing interests.
Figures

Similar articles
-
Dihydromyricetin improves DSS-induced colitis in mice via modulation of fecal-bacteria-related bile acid metabolism.Pharmacol Res. 2021 Sep;171:105767. doi: 10.1016/j.phrs.2021.105767. Epub 2021 Jul 14. Pharmacol Res. 2021. PMID: 34273490
-
Interleukin-15 promotes intestinal dysbiosis with butyrate deficiency associated with increased susceptibility to colitis.ISME J. 2017 Jan;11(1):15-30. doi: 10.1038/ismej.2016.114. Epub 2016 Sep 20. ISME J. 2017. PMID: 27648810 Free PMC article.
-
Fecal let-7b and miR-21 directly modulate the intestinal microbiota, driving chronic inflammation.Gut Microbes. 2024 Jan-Dec;16(1):2394249. doi: 10.1080/19490976.2024.2394249. Epub 2024 Sep 3. Gut Microbes. 2024. PMID: 39224018 Free PMC article.
-
Bridging intestinal immunity and gut microbiota by metabolites.Cell Mol Life Sci. 2019 Oct;76(20):3917-3937. doi: 10.1007/s00018-019-03190-6. Epub 2019 Jun 27. Cell Mol Life Sci. 2019. PMID: 31250035 Free PMC article. Review.
-
Impact of Bacterial Metabolites on Gut Barrier Function and Host Immunity: A Focus on Bacterial Metabolism and Its Relevance for Intestinal Inflammation.Front Immunol. 2021 May 26;12:658354. doi: 10.3389/fimmu.2021.658354. eCollection 2021. Front Immunol. 2021. PMID: 34122415 Free PMC article. Review.
Cited by
-
Designing synthetic microbial communities for enhanced anaerobic waste treatment.Appl Environ Microbiol. 2025 Jun 18;91(6):e0040425. doi: 10.1128/aem.00404-25. Epub 2025 May 16. Appl Environ Microbiol. 2025. PMID: 40377302 Free PMC article. Review.
-
Associations of Dietary Intake with the Intestinal Microbiota and Short-Chain Fatty Acids Among Young Adults with Type 1 Diabetes and Overweight or Obesity.J Nutr. 2023 Apr;153(4):1178-1188. doi: 10.1016/j.tjnut.2022.12.017. Epub 2022 Dec 27. J Nutr. 2023. PMID: 36841667 Free PMC article. Clinical Trial.
-
The gut microbiome dysbiosis and regulation by fecal microbiota transplantation: umbrella review.Front Microbiol. 2023 Nov 3;14:1286429. doi: 10.3389/fmicb.2023.1286429. eCollection 2023. Front Microbiol. 2023. PMID: 38029189 Free PMC article. Review.
-
Roseburia intestinalis and Its Metabolite Butyrate Inhibit Colitis and Upregulate TLR5 through the SP3 Signaling Pathway.Nutrients. 2022 Jul 25;14(15):3041. doi: 10.3390/nu14153041. Nutrients. 2022. PMID: 35893896 Free PMC article.
-
Bile Acid-Gut Microbiota Axis in Inflammatory Bowel Disease: From Bench to Bedside.Nutrients. 2021 Sep 9;13(9):3143. doi: 10.3390/nu13093143. Nutrients. 2021. PMID: 34579027 Free PMC article. Review.
References
-
- Eeckhaut V. et al. In 5th Probiotics, Prebiotics and New Foods Congress, Rome, Italy. http://www.probiotics-prebiotics-newfood.org/pdf/5th_Probiotics_Prebioti....
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

