Gut microbiome-host interactions in driving environmental pollutant trichloroethene-mediated autoimmunity
- PMID: 34051218
- PMCID: PMC8314436
- DOI: 10.1016/j.taap.2021.115597
Gut microbiome-host interactions in driving environmental pollutant trichloroethene-mediated autoimmunity
Abstract
Trichloroethene (TCE), a widely used industrial solvent, is associated with the development of autoimmune diseases (ADs), including systemic lupus erythematosus and autoimmune hepatitis. Increasing evidence support a linkage between altered gut microbiome composition and the onset of ADs. However, it is not clear how gut microbiome contributes to TCE-mediated autoimmunity, and initial triggers for microbiome-host interactions leading to systemic autoimmune responses remain unknown. To achieve this, female MRL+/+ mice were treated with 0.5 mg/ml TCE for 52 weeks and fecal samples were subjected to 16S rRNA sequencing to determine the microbiome composition. TCE exposure resulted in distinct bacterial community revealed by β-diversity analysis. Notably, we observed reduction in Lactobacillaceae, Rikenellaceae and Bifidobacteriaceae families, and enrichment of Akkermansiaceae and Lachnospiraceae families after TCE exposure. We also observed significantly increased colonic oxidative stress and inflammatory markers (CD14 and IL-1β), and decreased tight junction proteins (ZO-2, occludin and claudin-3). These changes were associated with increases in serum antinuclear and anti-smooth muscle antibodies and cytokines (IL-6 and IL-12), together with increased PD1 + CD4+ T cells in TCE-exposed spleen and liver tissues. Importantly, fecal microbiota transplantation (FMT) using feces from TCE-treated mice to antibiotics-treated mice induced increased anti-dsDNA antibodies and hepatic CD4+ T cell infiltration in the recipient mice. Our studies thus delineate how imbalance in gut microbiome and mucosal redox status together with gut inflammatory response and permeability changes could be the key factors in contributing to TCE-mediated ADs. Furthermore, FMT studies provide a solid support to a causal role of microbiome in TCE-mediated autoimmunity.
Keywords: Autoimmunity; Environmental pollutant; Inflammation; Microbiome; Oxidative stress; Permeability.
Published by Elsevier Inc.
Conflict of interest statement
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
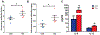




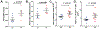
Similar articles
-
Aberrant Gut Microbiome Contributes to Intestinal Oxidative Stress, Barrier Dysfunction, Inflammation and Systemic Autoimmune Responses in MRL/lpr Mice.Front Immunol. 2021 Apr 12;12:651191. doi: 10.3389/fimmu.2021.651191. eCollection 2021. Front Immunol. 2021. PMID: 33912174 Free PMC article.
-
Redox regulation of hepatic NLRP3 inflammasome activation and immune dysregulation in trichloroethene-mediated autoimmunity.Free Radic Biol Med. 2019 Nov 1;143:223-231. doi: 10.1016/j.freeradbiomed.2019.08.014. Epub 2019 Aug 13. Free Radic Biol Med. 2019. PMID: 31419475 Free PMC article.
-
N-Acetylcysteine protects against trichloroethene-mediated autoimmunity by attenuating oxidative stress.Toxicol Appl Pharmacol. 2013 Nov 15;273(1):189-95. doi: 10.1016/j.taap.2013.08.020. Epub 2013 Aug 28. Toxicol Appl Pharmacol. 2013. PMID: 23993974 Free PMC article.
-
Evidence of autoimmune-related effects of trichloroethylene exposure from studies in mice and humans.Environ Health Perspect. 2009 May;117(5):696-702. doi: 10.1289/ehp.11782. Epub 2009 Jan 9. Environ Health Perspect. 2009. PMID: 19479009 Free PMC article. Review.
-
Environmental Exposures and Autoimmune Diseases: Contribution of Gut Microbiome.Front Immunol. 2020 Jan 10;10:3094. doi: 10.3389/fimmu.2019.03094. eCollection 2019. Front Immunol. 2020. PMID: 31998327 Free PMC article. Review.
Cited by
-
Oral ingestion of the environmental toxicant trichloroethylene in rats induces alterations in the gut microbiome: Relevance to idiopathic Parkinson's disease.Toxicol Appl Pharmacol. 2022 Sep 15;451:116176. doi: 10.1016/j.taap.2022.116176. Epub 2022 Jul 29. Toxicol Appl Pharmacol. 2022. PMID: 35914559 Free PMC article.
-
Effects of Probiotics on Liver Diseases: Current In Vitro and In Vivo Studies.Probiotics Antimicrob Proteins. 2025 Jun;17(3):1688-1710. doi: 10.1007/s12602-024-10431-z. Epub 2024 Dec 30. Probiotics Antimicrob Proteins. 2025. PMID: 39739162 Review.
-
Interacting roles of gut microbiota and T cells in the development of autoimmune hepatitis.Front Immunol. 2025 May 26;16:1584001. doi: 10.3389/fimmu.2025.1584001. eCollection 2025. Front Immunol. 2025. PMID: 40491914 Free PMC article. Review.
-
Trichloroethylene: An Update on an Environmental Contaminant with Multiple Health Effects.Annu Rev Pharmacol Toxicol. 2025 Jan;65(1):507-527. doi: 10.1146/annurev-pharmtox-022724-120525. Epub 2024 Dec 17. Annu Rev Pharmacol Toxicol. 2025. PMID: 39094062 Free PMC article. Review.
-
Role of Intestinal Microbes in Chronic Liver Diseases.Int J Mol Sci. 2022 Oct 21;23(20):12661. doi: 10.3390/ijms232012661. Int J Mol Sci. 2022. PMID: 36293518 Free PMC article. Review.
References
-
- Albillos A, de Gottardi A, Rescigno M, 2020. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol 72, 558–577. - PubMed
-
- Anagnostopoulos G, Sakorafas GH, Grigoriadis K, Margantinis G, Kostopoulos P, Tsiakos S, Arvanitidis D, 2004. Hepatitis caused by occupational chronic exposure to trichloroethylene. Acta Gastroenterol Belg 67, 355–357. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

