JAK-STAT in Early Hematopoiesis and Leukemia
- PMID: 34055801
- PMCID: PMC8160090
- DOI: 10.3389/fcell.2021.669363
JAK-STAT in Early Hematopoiesis and Leukemia
Abstract
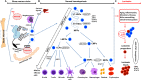
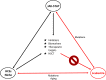
Hematopoietic stem cells (HSCs) produce all the terminally differentiated blood cells and are controlled by extracellular signals from the microenvironment, the bone marrow (BM) niche, as well as intrinsic cell signals. Intrinsic signals include the tightly controlled action of signaling pathways, as the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway. Activation of JAK-STAT leads to phosphorylation of members of the STAT family to regulate proliferation, survival, and self-renewal of HSCs. Mutations in components of the JAK-STAT pathway are linked with defects in HSCs and hematologic malignancies. Accumulating mutations in HSCs and aging contribute to leukemia transformation. Here an overview of hematopoiesis, and the role of the JAK-STAT pathway in HSCs and in the promotion of leukemic transformation is presented. Therapeutic targeting of JAK-STAT and clinical implications of the existing research findings are also discussed.
Keywords: JAK-STAT; STAT5; STATs; hematopoiesis; hematopoietic stem cells; leukemia.
Copyright © 2021 Fasouli and Katsantoni.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Baccin C., Al-Sabah J., Velten L., Helbling P. M., Grunschlager F., Hernandez-Malmierca P., et al. (2020). Combined single-cell and spatial transcriptomics reveal the molecular, cellular and spatial bone marrow niche organization. Nat. Cell Biol. 22 38–48. 10.1038/s41556-019-0439-6 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

