Gold Nanoparticle-Bioconjugated Aminoguanidine Inhibits Glycation Reaction: An In Vivo Study in a Diabetic Animal Model
- PMID: 34055984
- PMCID: PMC8137289
- DOI: 10.1155/2021/5591851
Gold Nanoparticle-Bioconjugated Aminoguanidine Inhibits Glycation Reaction: An In Vivo Study in a Diabetic Animal Model
Abstract
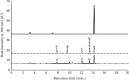
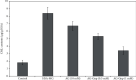
Proteins undergo glycation resulting in the generation of advanced glycation end products (AGEs) that play a central role in the onset and advancement of diabetes-associated secondary complications. Aminoguanidine (AG) acts as an antiglycating agent by inhibiting AGE generation by blocking reactive carbonyl species (RCS) like, methylglyoxal (MGO). Previous studies on antiglycating behavior of AG gave promising results in the treatment of diabetes-associated microvascular complications, but it was discontinued as it was found to be toxic at high concentrations (>10 mmol/L). The current article aims at glycation inhibition by conjugating gold nanoparticles (Gnp) with less concentration of AG (0.5-1.0 mmol/L). The HPLC results showed that AG-Gnp fairly hampers the formation of glycation adducts. Moreover, the in vivo studies revealed AG-Gnp mediated inhibition in the production of total-AGEs and -N ε -(carboxymethyl)lysine (CML) in the diabetic rat model. This inhibition was found to be directly correlated with the antioxidant parameters, blood glucose, insulin, and glycosylated hemoglobin levels. Furthermore, the histopathology of AG-Gnp-treated rats showed good recovery in the damaged pancreatic tissue as compared to diabetic rats. We propose that this approach might increase the efficacy of AG at relatively low concentrations to avoid toxicity and might facilitate to overcome the hazardous actions of antiglycating drugs.
Copyright © 2021 Saheem Ahmad et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

