Simultaneous Pharmacologic Inhibition of Yes-Associated Protein 1 and Glutaminase 1 via Inhaled Poly(Lactic-co-Glycolic) Acid-Encapsulated Microparticles Improves Pulmonary Hypertension
- PMID: 34056915
- PMCID: PMC8477870
- DOI: 10.1161/JAHA.120.019091
Simultaneous Pharmacologic Inhibition of Yes-Associated Protein 1 and Glutaminase 1 via Inhaled Poly(Lactic-co-Glycolic) Acid-Encapsulated Microparticles Improves Pulmonary Hypertension
Abstract
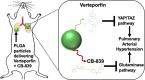
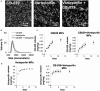
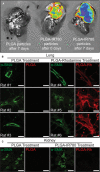
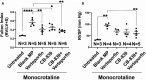
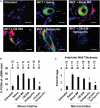
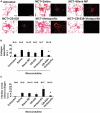
Background Pulmonary hypertension (PH) is a deadly disease characterized by vascular stiffness and altered cellular metabolism. Current treatments focus on vasodilation and not other root causes of pathogenesis. Previously, it was demonstrated that glutamine metabolism, as catalyzed by GLS1 (glutaminase 1) activity, is mechanoactivated by matrix stiffening and the transcriptional coactivators YAP1 (yes-associated protein 1) and transcriptional coactivator with PDZ-binding motif (TAZ), resulting in pulmonary vascular proliferation and PH. Pharmacologic inhibition of YAP1 (by verteporfin) or glutaminase (by CB-839) improved PH in vivo. However, systemic delivery of these agents, particularly YAP1 inhibitors, may have adverse chronic effects. Furthermore, simultaneous use of pharmacologic blockers may offer additive or synergistic benefits. Therefore, a strategy that delivers these drugs in combination to local lung tissue, thus avoiding systemic toxicity and driving more robust improvement, was investigated. Methods and Results We used poly(lactic-co-glycolic) acid polymer-based microparticles for delivery of verteporfin and CB-839 simultaneously to the lungs of rats suffering from monocrotaline-induced PH. Microparticles released these drugs in a sustained fashion and delivered their payload in the lungs for 7 days. When given orotracheally to the rats weekly for 3 weeks, microparticles carrying this drug combination improved hemodynamic (right ventricular systolic pressure and right ventricle/left ventricle+septum mass ratio), histologic (vascular remodeling), and molecular markers (vascular proliferation and stiffening) of PH. Importantly, only the combination of drug delivery, but neither verteporfin nor CB-839 alone, displayed significant improvement across all indexes of PH. Conclusions Simultaneous, lung-specific, and controlled release of drugs targeting YAP1 and GLS1 improved PH in rats, addressing unmet needs for the treatment of this deadly disease.
Keywords: mechanotransduction; metabolism; nanoparticle; pulmonary hypertension; therapy.
Conflict of interest statement
S.Y.C. has served as a consultant for Zogenix and United Therapeutics and has held grants from Pfizer and Actelion. S.Y.C. serves as a founder, director, and officer of Synhale Therapeutics. Patent applications (S.Y.C., T.B., A.P.A., S.R.L.) have been filed regarding targeting YAP1 (yes‐associated protein 1)/TAZ (transcriptional coactivator with PDZ‐binding motif), glutaminase, and glutamine metabolism in pulmonary hypertension. The remaining authors have no disclosures to report.
Figures

References
-
- Dumas SJ, Bru‐Mercier G, Courboulin A, Quatredeniers M, Rücker‐Martin C, Antigny F, Nakhleh MK, Ranchoux B, Gouadon E, Vinhas M‐C, et al. NMDA‐type glutamate receptor activation promotes vascular remodeling and pulmonary arterial hypertension. Circulation. 2018;137:2371–2389. DOI: 10.1161/CIRCULATIONAHA.117.029930. - DOI - PubMed
-
- Bertero T, Cottrill K, Lu YU, Haeger C, Dieffenbach P, Annis S, Hale A, Bhat B, Kaimal V, Zhang Y‐Y, et al. Matrix remodeling promotes pulmonary hypertension through feedback mechanoactivation of the YAP/TAZ‐miR‐130/301 circuit. Cell Rep. 2015;13:1016–1032. DOI: 10.1016/j.celrep.2015.09.049. - DOI - PMC - PubMed
-
- Kudryashova TV, Goncharov DA, Pena A, Kelly N, Vanderpool R, Baust J, Kobir A, Shufesky W, Mora AL, Morelli AE, et al. Hippo‐integrin‐linked kinase cross‐talk controls self‐sustaining proliferation and survival in pulmonary hypertension. Am J Respir Crit Care Med. 2016;194:866–877. DOI: 10.1164/rccm.201510-2003OC. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

