High-throughput PBTK models for in vitro to in vivo extrapolation
- PMID: 34056988
- PMCID: PMC9703392
- DOI: 10.1080/17425255.2021.1935867
High-throughput PBTK models for in vitro to in vivo extrapolation
Abstract
Introduction: Toxicity data are unavailable for many thousands of chemicals in commerce and the environment. Therefore, risk assessors need to rapidly screen these chemicals for potential risk to public health. High-throughput screening (HTS) for in vitro bioactivity, when used with high-throughput toxicokinetic (HTTK) data and models, allows characterization of these thousands of chemicals.
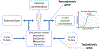
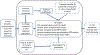
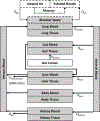
Areas covered: This review covers generic physiologically based toxicokinetic (PBTK) models and high-throughput PBTK modeling for in vitro-in vivo extrapolation (IVIVE) of HTS data. We focus on 'httk', a public, open-source set of computational modeling tools and in vitro toxicokinetic (TK) data.
Expert opinion: HTTK benefits chemical risk assessors with its ability to support rapid chemical screening/prioritization, perform IVIVE, and provide provisional TK modeling for large numbers of chemicals using only limited chemical-specific data. Although generic TK model design can increase prediction uncertainty, these models provide offsetting benefits by increasing model implementation accuracy. Also, public distribution of the models and data enhances reproducibility. For the httk package, the modular and open-source design can enable the tool to be used and continuously improved by a broad user community in support of the critical need for high-throughput chemical prioritization and rapid dose estimation to facilitate rapid hazard assessments.
Keywords: Generic physiologically based toxicokinetic models; high-throughput; in vitro to in vivo extrapolation; modeling software tools; open source tools; physiologically-based toxicokinetics; toxicokinetics.
Figures

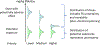
References
-
- Rager JE, Strynar MJ, Liang S, et al. Linking high resolution mass spectrometry data with exposure and toxicity forecasts to advance high-throughput environmental monitoring. Environment International. 2016;88:269–280. - PubMed
-
- Egeghy PP, Judson R, Gangwal S, et al. The exposure data landscape for manufactured chemicals. Science of the Total Environment. 2012. Jan 1;414:159–66. - PubMed
-
- Breyer S. Breaking the Vicious Circle: Toward Effective Risk Regulation. Harvard University Press; 2009.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
