Immune exhaustion in chronic Chagas disease: Pro-inflammatory and immunomodulatory action of IL-27 in vitro
- PMID: 34061845
- PMCID: PMC8195349
- DOI: 10.1371/journal.pntd.0009473
Immune exhaustion in chronic Chagas disease: Pro-inflammatory and immunomodulatory action of IL-27 in vitro
Abstract
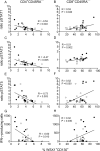
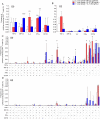
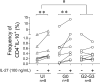
In chronic Chagas disease, Trypanosoma cruzi-specific T-cell function decreases over time, and alterations in the homeostatic IL-7/IL-7R axis are evident, consistent with a process of immune exhaustion. IL-27 is an important immunoregulatory cytokine that shares T-cell signaling with IL-7 and other cytokines of the IL-12 family and might be involved in the transcriptional regulation of T-cell function. Here, we evaluated the expression and function of IL-27R in antigen-experienced T cells from subjects with chronic Chagas disease and assessed whether in vitro treatment with IL-27 and IL-7 might improve T. cruzi-specific polyfunctional T-cell responses. In vitro exposure of PBMCs to T. cruzi induced a downregulation of IL-27R in CD4+ T cells and an upregulation in CD8+ T cells in subjects without heart disease, while IL-27R expression remained unaltered in subjects with more severe clinical stages. The modulation of IL-27R was associated with functional signaling through STAT3 and STAT5 and induction of the downstream genes TBX21, EOMES and CXCL9 in response to IL-27. In vitro treatment of PBMCs with IL-27 and IL-7 improved monofunctional and polyfunctional Th1 responses, accompanied by the induction of IL-10 and Bcl-2 expression in subjects without heart disease but did not improve those in subjects with cardiomyopathy. Our findings support the process of desensitization of the IL-27/IL-27R pathway along with disease severity and that the pro-inflammatory and immunomodulatory mechanisms of IL-27 might be interconnected.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
Trypanosoma cruzi-specific IFN-γ-producing cells in chronic Chagas disease associate with a functional IL-7/IL-7R axis.PLoS Negl Trop Dis. 2018 Dec 5;12(12):e0006998. doi: 10.1371/journal.pntd.0006998. eCollection 2018 Dec. PLoS Negl Trop Dis. 2018. PMID: 30517089 Free PMC article.
-
Perturbed T cell IL-7 receptor signaling in chronic Chagas disease.J Immunol. 2015 Apr 15;194(8):3883-9. doi: 10.4049/jimmunol.1402202. Epub 2015 Mar 13. J Immunol. 2015. PMID: 25769928 Free PMC article. Clinical Trial.
-
Impact of benznidazole treatment on the functional response of Trypanosoma cruzi antigen-specific CD4+CD8+ T cells in chronic Chagas disease patients.PLoS Negl Trop Dis. 2018 May 11;12(5):e0006480. doi: 10.1371/journal.pntd.0006480. eCollection 2018 May. PLoS Negl Trop Dis. 2018. PMID: 29750791 Free PMC article.
-
[Role of cytokines in resistance and pathology in Trypanosoma cruzi infection].Rev Argent Microbiol. 1996 Apr-Jun;28(2):99-109. Rev Argent Microbiol. 1996. PMID: 8768488 Review. Spanish.
-
Generation, specificity, and function of CD8+ T cells in Trypanosoma cruzi infection.Immunol Rev. 2004 Oct;201:304-17. doi: 10.1111/j.0105-2896.2004.00183.x. Immunol Rev. 2004. PMID: 15361249 Review.
Cited by
-
Chagas disease in immunocompromised patients.Clin Microbiol Rev. 2024 Jun 13;37(2):e0009923. doi: 10.1128/cmr.00099-23. Epub 2024 Mar 28. Clin Microbiol Rev. 2024. PMID: 38546225 Free PMC article. Review.
-
C57BL/6 Mice Pretreated With Alpha-Tocopherol Show a Better Outcome of Trypanosoma cruzi Infection With Less Tissue Inflammation and Fibrosis.Front Immunol. 2022 Jan 28;13:833560. doi: 10.3389/fimmu.2022.833560. eCollection 2022. Front Immunol. 2022. PMID: 35154155 Free PMC article.
-
The Functions of Cytokines in the Cardiac Immunopathogenesis of Chagas Disease.Pathogens. 2024 Oct 3;13(10):870. doi: 10.3390/pathogens13100870. Pathogens. 2024. PMID: 39452741 Free PMC article. Review.
-
Intra-host Trypanosoma cruzi strain dynamics shape disease progression: the missing link in Chagas disease pathogenesis.Microbiol Spectr. 2023 Sep 5;11(5):e0423622. doi: 10.1128/spectrum.04236-22. Online ahead of print. Microbiol Spectr. 2023. PMID: 37668388 Free PMC article.
References
-
- WHO. Fact sheet: Chagas disease (American trypanosomiasis) [Internet]. 2020 p. who.int/mediacentre/factsheets/fs340/en/. Available: who.int/mediacentre/factsheets/fs340/en/
-
- Dutra WO, Menezes CAS, Villani FNA, da Costa GC, da Silveira ABM, Reis DDÁ, et al.. Cellular and genetic mechanisms involved in the generation of protective and pathogenic immune responses in human Chagas disease. Mem Inst Oswaldo Cruz. 2009;104: 208–218. doi: 10.1590/s0074-02762009000900027 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

