The Zebrafish Cardiac Endothelial Cell-Roles in Development and Regeneration
- PMID: 34062899
- PMCID: PMC8147271
- DOI: 10.3390/jcdd8050049
The Zebrafish Cardiac Endothelial Cell-Roles in Development and Regeneration
Abstract
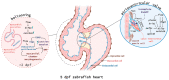
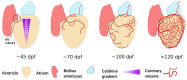
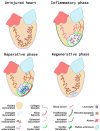
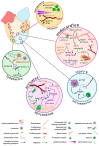
In zebrafish, the spatiotemporal development of the vascular system is well described due to its stereotypical nature. However, the cellular and molecular mechanisms orchestrating post-embryonic vascular development, the maintenance of vascular homeostasis, or how coronary vessels integrate into the growing heart are less well studied. In the context of cardiac regeneration, the central cellular mechanism by which the heart regenerates a fully functional myocardium relies on the proliferation of pre-existing cardiomyocytes; the epicardium and the endocardium are also known to play key roles in the regenerative process. Remarkably, revascularisation of the injured tissue occurs within a few hours after cardiac damage, thus generating a vascular network acting as a scaffold for the regenerating myocardium. The activation of the endocardium leads to the secretion of cytokines, further supporting the proliferation of the cardiomyocytes. Although epicardium, endocardium, and myocardium interact with each other to orchestrate heart development and regeneration, in this review, we focus on recent advances in the understanding of the development of the endocardium and the coronary vasculature in zebrafish as well as their pivotal roles in the heart regeneration process.
Keywords: coronary vessels; development; endocardium; heart; lymphatics; regeneration; zebrafish.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Krauss J., Astrinidis P., Astrinides P., Frohnhöfer H.G., Walderich B., Nüsslein-Volhard C. Transparent, a gene affecting stripe formation in Zebrafish, encodes the mitochondrial protein Mpv17 that is required for iridophore survival. Biol. Open. 2013;2:703–710. doi: 10.1242/bio.20135132. - DOI - PMC - PubMed
-
- White A.J., Boffa M.B., Jones B., Petkovich M. A zebrafish retinoic acid receptor expressed in the regenerating caudal fin. Developement. 1994;120:1861–1872. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

