Microvascular Tissue Engineering-A Review
- PMID: 34064101
- PMCID: PMC8224375
- DOI: 10.3390/biomedicines9060589
Microvascular Tissue Engineering-A Review
Abstract
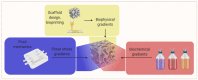
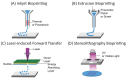
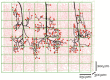
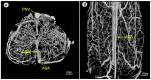
Tissue engineering and regenerative medicine have come a long way in recent decades, but the lack of functioning vasculature is still a major obstacle preventing the development of thicker, physiologically relevant tissue constructs. A large part of this obstacle lies in the development of the vessels on a microscale-the microvasculature-that are crucial for oxygen and nutrient delivery. In this review, we present the state of the art in the field of microvascular tissue engineering and demonstrate the challenges for future research in various sections of the field. Finally, we illustrate the potential strategies for addressing some of those challenges.
Keywords: biomaterials; coculture; gradients; microvascularization; regenerative medicine; tissue engineering; vascularization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- Brey E. Vascularization: Regenerative Medicine and Tissue Engineering. Volume 1 Taylor & Francis Group, LLC; Boca Raton, FL, USA: 2015.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

