Targeting the NLRP3 Inflammasome as a New Therapeutic Option for Overcoming Cancer
- PMID: 34064909
- PMCID: PMC8151587
- DOI: 10.3390/cancers13102297
Targeting the NLRP3 Inflammasome as a New Therapeutic Option for Overcoming Cancer
Abstract
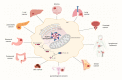
Inflammasomes are multiprotein complexes that regulate the maturation and secretion of the proinflammatory cytokines interleukin-1beta (IL-1β and interleukin-18 (IL-18) in response to various intracellular stimuli. As a member of the inflammasomes family, NLRP3 is the most studied and best characterized inflammasome and has been shown to be involved in several pathologies. Recent findings have made it increasingly apparent that the NLRP3 inflammasome may also play a central role in tumorigenesis, and it has attracted attention as a potential anticancer therapy target. In this review, we discuss the role of NLRP3 in the development and progression of cancer, offering a detailed summary of NLRP3 inflammasome activation (and inhibition) in the pathogenesis of various forms of cancer. Moreover, we focus on the therapeutic potential of targeting NLRP3 for cancer therapy, emphasizing how understanding NLRP3 inflammasome-dependent cancer mechanisms might guide the development of new drugs that target the inflammatory response of tumor-associated cells.
Keywords: NLRP3 inflammasome; inflammation; inhibitors; target-therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
The NLRP3 inflammasome: a potential therapeutic target for traumatic brain injury.Neural Regen Res. 2021 Jan;16(1):49-57. doi: 10.4103/1673-5374.286951. Neural Regen Res. 2021. PMID: 32788447 Free PMC article.
-
Cyclooxygenase-2 regulates NLRP3 inflammasome-derived IL-1β production.J Cell Physiol. 2015 Apr;230(4):863-74. doi: 10.1002/jcp.24815. J Cell Physiol. 2015. PMID: 25294243
-
Role of NLRP3 inflammasome in inflammatory bowel diseases.World J Gastroenterol. 2019 Sep 7;25(33):4796-4804. doi: 10.3748/wjg.v25.i33.4796. World J Gastroenterol. 2019. PMID: 31543674 Free PMC article.
-
Recent Advances of the NLRP3 Inflammasome in Central Nervous System Disorders.J Immunol Res. 2016;2016:9238290. doi: 10.1155/2016/9238290. Epub 2016 Aug 29. J Immunol Res. 2016. PMID: 27652274 Free PMC article. Review.
-
Therapeutic Targeting of NLRP3 Inflammasomes by Natural Products and Pharmaceuticals: A Novel Mechanistic Approach for Inflammatory Diseases.Curr Med Chem. 2017;24(16):1645-1670. doi: 10.2174/0929867324666170227121619. Curr Med Chem. 2017. PMID: 28245768 Review.
Cited by
-
Immunomodulatory Role of Thioredoxin Interacting Protein in Cancer's Impediments: Current Understanding and Therapeutic Implications.Vaccines (Basel). 2022 Nov 10;10(11):1902. doi: 10.3390/vaccines10111902. Vaccines (Basel). 2022. PMID: 36366411 Free PMC article. Review.
-
Dexamethasone and OLT1177 Cooperate in the Reduction of Melanoma Growth by Inhibiting STAT3 Functions.Cells. 2023 Jan 12;12(2):294. doi: 10.3390/cells12020294. Cells. 2023. PMID: 36672229 Free PMC article.
-
Synthesis and NLRP3-Inflammasome Inhibitory Activity of the Naturally Occurring Velutone F and of Its Non-Natural Regioisomeric Chalconoids.Int J Mol Sci. 2022 Aug 11;23(16):8957. doi: 10.3390/ijms23168957. Int J Mol Sci. 2022. PMID: 36012241 Free PMC article.
-
Decreased survival in patients with pancreatic cancer may be associated with an increase in histopathological expression of inflammasome marker NLRP3.Histol Histopathol. 2024 Jan;39(1):35-40. doi: 10.14670/HH-18-617. Epub 2023 Apr 4. Histol Histopathol. 2024. PMID: 37057822
-
Amino Sugar-Enriched Fraction of Korean Red Ginseng Extract Induces the Priming Step of NLRP3 Inflammasome.Molecules. 2024 Mar 24;29(7):1455. doi: 10.3390/molecules29071455. Molecules. 2024. PMID: 38611734 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

