Peptide-Based Electrospun Fibers: Current Status and Emerging Developments
- PMID: 34065019
- PMCID: PMC8151459
- DOI: 10.3390/nano11051262
Peptide-Based Electrospun Fibers: Current Status and Emerging Developments
Abstract
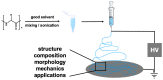
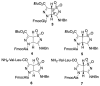
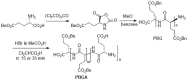
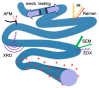
Electrospinning is a well-known, straightforward, and versatile technique, widely used for the preparation of fibers by electrifying a polymer solution. However, a high molecular weight is not essential for obtaining uniform electrospun fibers; in fact, the primary criterion to succeed is the presence of sufficient intermolecular interactions, which function similar to chain entanglements. Some small molecules able to self-assemble have been electrospun from solution into fibers and, among them, peptides containing both natural and non-natural amino acids are of particular relevance. Nowadays, the use of peptides for this purpose is at an early stage, but it is gaining more and more interest, and we are now witnessing the transition from basic research towards applications. Considering the novelty in the relevant processing, the aim of this review is to analyze the state of the art from the early 2000s on. Moreover, advantages and drawbacks in using peptides as the main or sole component for generating electrospun nanofibers will be discussed. Characterization techniques that are specifically targeted to the produced peptide fibers are presented.
Keywords: electrospinning; peptide-based electrospun nanofibers; peptides; peptidomimetics; self-assembly.
Conflict of interest statement
The authors declare no conflict of interest.
Figures










References
-
- Reneker D.H., Yarin A.L. Electrospinning jets and polymer nanofibers. Polymer. 2008;49:2387–2425. doi: 10.1016/j.polymer.2008.02.002. - DOI
-
- Singer J.C., Ringk A., Giesa R., Schmidt H.-W. Melt Electrospinning of Small Molecules. Macromol. Mater. Eng. 2015;300:259–276. doi: 10.1002/mame.201400296. - DOI
Publication types
LinkOut - more resources
Full Text Sources

