5-Aminolevulinic Acid as a Novel Therapeutic for Inflammatory Bowel Disease
- PMID: 34065300
- PMCID: PMC8160866
- DOI: 10.3390/biomedicines9050578
5-Aminolevulinic Acid as a Novel Therapeutic for Inflammatory Bowel Disease
Abstract
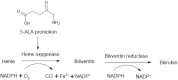
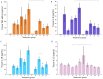
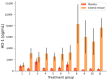
5-Aminolevulinic acid (5-ALA) is a naturally occurring nonprotein amino acid licensed as an optical imaging agent for the treatment of gliomas. In recent years, 5-ALA has been shown to possess anti-inflammatory and immunoregulatory properties through upregulation of heme oxygenase-1 via enhancement of porphyrin, indicating that it may be beneficial for the treatment of inflammatory conditions. This study systematically examines 5-ALA for use in inflammatory bowel disease (IBD). Firstly, the ex vivo colonic stability and permeability of 5-ALA was assessed using human and mouse fluid and tissue. Secondly, the in vivo efficacy of 5-ALA, in the presence of sodium ferrous citrate, was investigated via the oral and intracolonic route in an acute DSS colitis mouse model of IBD. Results showed that 5-ALA was stable in mouse and human colon fluid, as well as in colon tissue. 5-ALA showed more tissue restricted pharmacokinetics when exposed to human colonic tissue. In vivo dosing demonstrated significantly improved colonic inflammation, increased local heme oxygenase-1 levels, and decreased concentrations of proinflammatory cytokines TNF-α, IL-6, and IL-1β in both plasma and colonic tissue. These effects were superior to that measured concurrently with established anti-inflammatory treatments, ciclosporin and 5-aminosalicylic acid (mesalazine). As such, 5-ALA represents a promising addition to the IBD armamentarium, with potential for targeted colonic delivery.
Keywords: 5 amino levulinic acid; Crohn’s disease; anti-inflammatory; colonic drug delivery; drug stability; inflammation; large intestine; microbiome; microbiota metabolism; ulcerative colitis.
Conflict of interest statement
The authors declare no other conflict of interest than the patent mentioned above. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

Similar articles
-
Ursodeoxycholic acid and lithocholic acid exert anti-inflammatory actions in the colon.Am J Physiol Gastrointest Liver Physiol. 2017 Jun 1;312(6):G550-G558. doi: 10.1152/ajpgi.00256.2016. Epub 2017 Mar 30. Am J Physiol Gastrointest Liver Physiol. 2017. PMID: 28360029
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
-
Anti-inflammatory action of a novel orally available peptide 317 in mouse models of inflammatory bowel diseases.Pharmacol Rep. 2014 Oct;66(5):741-50. doi: 10.1016/j.pharep.2014.03.007. Epub 2014 Apr 4. Pharmacol Rep. 2014. PMID: 25149976
-
Attenuation of colonic inflammation by partial replacement of dietary linoleic acid with α-linolenic acid in a rat model of inflammatory bowel disease.Br J Nutr. 2012 Nov 14;108(9):1612-22. doi: 10.1017/S0007114511007197. Epub 2012 Jan 16. Br J Nutr. 2012. PMID: 22243775
-
5-Aminolevulinic acid regulates the inflammatory response and alloimmune reaction.Int Immunopharmacol. 2016 Aug;37:71-78. doi: 10.1016/j.intimp.2015.11.034. Epub 2015 Nov 28. Int Immunopharmacol. 2016. PMID: 26643355 Review.
Cited by
-
The diagnostic potential of urine in paediatric patients undergoing initial treatment for tuberculous meningitis.Sci Rep. 2024 Aug 22;14(1):19471. doi: 10.1038/s41598-024-70419-1. Sci Rep. 2024. PMID: 39174657 Free PMC article.
-
Dual action tofacitinib-loaded PLGA nanoparticles alleviate colitis in an IBD mouse model.Drug Deliv Transl Res. 2025 Jul;15(7):2372-2389. doi: 10.1007/s13346-024-01736-1. Epub 2024 Nov 11. Drug Deliv Transl Res. 2025. PMID: 39527394 Free PMC article.
-
Ileocolonic-Targeted JAK Inhibitor: A Safer and More Effective Treatment for Inflammatory Bowel Disease.Pharmaceutics. 2022 Nov 5;14(11):2385. doi: 10.3390/pharmaceutics14112385. Pharmaceutics. 2022. PMID: 36365202 Free PMC article.
-
Machine Learning Predicts Drug Metabolism and Bioaccumulation by Intestinal Microbiota.Pharmaceutics. 2021 Nov 25;13(12):2001. doi: 10.3390/pharmaceutics13122001. Pharmaceutics. 2021. PMID: 34959282 Free PMC article.
-
Blood-Based Immune Protein Markers of Disease Progression in Murine Models of Acute and Chronic Inflammatory Bowel Disease.Biomedicines. 2023 Jan 5;11(1):140. doi: 10.3390/biomedicines11010140. Biomedicines. 2023. PMID: 36672648 Free PMC article.
References
-
- Alatab S., Sepanlou S.G., Ikuta K., Vahedi H., Bisignano C., Safiri S., Sadeghi A., Nixon M.R., Abdoli A., Abolhassani H., et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study. Lancet Gastroenterol. Hepatol. 2020;5:17–30. doi: 10.1016/S2468-1253(19)30333-4. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

