Gut Organoid as a New Platform to Study Alginate and Chitosan Mediated PLGA Nanoparticles for Drug Delivery
- PMID: 34065505
- PMCID: PMC8161322
- DOI: 10.3390/md19050282
Gut Organoid as a New Platform to Study Alginate and Chitosan Mediated PLGA Nanoparticles for Drug Delivery
Abstract
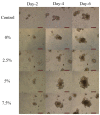
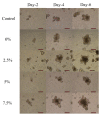
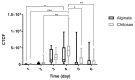
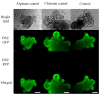
Intestinal organoids can be used as an ex vivo epithelial model to study different drug delivery effects on epithelial cells' luminal surface. In this study, the impact of surface charge on the delivery of 5-ASA loaded PLGA nanoparticles into the lumen of organoids was investigated. Alginate and chitosan were used to coat the nanoparticles and provide negative and positive charges on the particles, respectively. The organoid growth and viability were not affected by the presence of either alginate- or chitosan-coated nanoparticles. It was shown that nanoparticles could be transported from the serosal side of the organoids to the lumen as the dye gradually accumulated in the lumen by day 2-3 after adding the nanoparticles to the Matrigel. By day 5, the dye was eliminated from the lumen of the organoids. It was concluded that the positively charged nanoparticles were more readily transported across the epithelium into the lumen. It may be attributed to the affinity of epithelial cells to the positive charge. Thus, the organoid can be utilized as an appropriate model to mimic the functions of the intestinal epithelium and can be used as a model to evaluate the benefits of nanoparticle-based drug delivery.
Keywords: PLGA; alginate; chitosan; nanoparticles; organoids.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the study’s design, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Figures






Similar articles
-
Biodegradable Nanoparticles-Loaded PLGA Microcapsule for the Enhanced Encapsulation Efficiency and Controlled Release of Hydrophilic Drug.Int J Mol Sci. 2021 Mar 10;22(6):2792. doi: 10.3390/ijms22062792. Int J Mol Sci. 2021. PMID: 33801871 Free PMC article.
-
Intestinal organoids containing poly(lactic-co-glycolic acid) nanoparticles for the treatment of inflammatory bowel diseases.J Biomed Mater Res A. 2018 Apr;106(4):876-886. doi: 10.1002/jbm.a.36305. Epub 2017 Dec 21. J Biomed Mater Res A. 2018. PMID: 29226615 Free PMC article.
-
Preparation, characterization, and in vitro and in vivo investigation of chitosan-coated poly (d,l-lactide-co-glycolide) nanoparticles for intestinal delivery of exendin-4.Int J Nanomedicine. 2013;8:1141-54. doi: 10.2147/IJN.S41457. Epub 2013 Mar 15. Int J Nanomedicine. 2013. PMID: 23658482 Free PMC article.
-
Chitosan/Alginate-Based Nanoparticles for Antibacterial Agents Delivery.Int J Nanomedicine. 2024 May 30;19:5021-5044. doi: 10.2147/IJN.S469572. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38832335 Free PMC article. Review.
-
Intestinal organoid cocultures with microbes.Nat Protoc. 2021 Oct;16(10):4633-4649. doi: 10.1038/s41596-021-00589-z. Epub 2021 Aug 11. Nat Protoc. 2021. PMID: 34381208 Review.
Cited by
-
Preparation of chitosan-coated hollow tin dioxide nanoparticles and their application in improving the oral bioavailability of febuxostat.Int J Pharm X. 2023 Jul 13;6:100199. doi: 10.1016/j.ijpx.2023.100199. eCollection 2023 Dec 15. Int J Pharm X. 2023. PMID: 37521247 Free PMC article.
-
Multifunctional sponge scaffold loaded with concentrated growth factors for promoting wound healing.iScience. 2022 Dec 20;26(1):105835. doi: 10.1016/j.isci.2022.105835. eCollection 2023 Jan 20. iScience. 2022. PMID: 36624841 Free PMC article.
-
Organoids in gastrointestinal diseases: from bench to clinic.MedComm (2020). 2024 Jun 29;5(7):e574. doi: 10.1002/mco2.574. eCollection 2024 Jul. MedComm (2020). 2024. PMID: 38948115 Free PMC article. Review.
-
Engineered organoids in oral and maxillofacial regeneration.iScience. 2022 Dec 7;26(1):105757. doi: 10.1016/j.isci.2022.105757. eCollection 2023 Jan 20. iScience. 2022. PMID: 36590157 Free PMC article. Review.
-
Chitosan-Poly(Acrylic Acid) Nanoparticles Loaded with R848 and MnCl2 Inhibit Melanoma via Regulating Macrophage Polarization and Dendritic Cell Maturation.Int J Nanomedicine. 2021 Aug 21;16:5675-5692. doi: 10.2147/IJN.S318363. eCollection 2021. Int J Nanomedicine. 2021. PMID: 34456564 Free PMC article.
References
-
- Michalčíková R.B., Dryahina K., Španěl P. SIFT-MS quantification of several breath biomarkers of inflammatory bowel disease, IBD: A detailed study of the ion chemistry. Int. J. Mass Spectrom. 2016;396:35–41. doi: 10.1016/j.ijms.2015.12.007. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

