Synthesis, Characterizations, and 9.4 Tesla T2 MR Images of Polyacrylic Acid-Coated Terbium(III) and Holmium(III) Oxide Nanoparticles
- PMID: 34065511
- PMCID: PMC8160651
- DOI: 10.3390/nano11051355
Synthesis, Characterizations, and 9.4 Tesla T2 MR Images of Polyacrylic Acid-Coated Terbium(III) and Holmium(III) Oxide Nanoparticles
Abstract
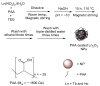
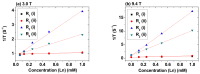
Polyacrylic acid (PAA)-coated lanthanide oxide (Ln2O3) nanoparticles (NPs) (Ln = Tb and Ho) with high colloidal stability and good biocompatibility were synthesized, characterized, and investigated as a new class of negative (T2) magnetic resonance imaging (MRI) contrast agents at high MR fields. Their r2 values were appreciable at a 3.0 T MR field and higher at a 9.4 T MR field, whereas their r1 values were negligible at all MR fields, indicating their exclusive induction of T2 relaxations with negligible induction of T1 relaxations. Their effectiveness as T2 MRI contrast agents at high MR fields was confirmed from strong negative contrast enhancements in in vivo T2 MR images at a 9.4 T MR field after intravenous administration into mice tails.
Keywords: Ho2O3; MRI; Tb2O3; high MR field; nanoparticles; polyacrylic acid-coating.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

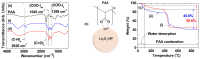




Similar articles
-
Functionalized Lanthanide Oxide Nanoparticles for Tumor Targeting, Medical Imaging, and Therapy.Pharmaceutics. 2021 Nov 8;13(11):1890. doi: 10.3390/pharmaceutics13111890. Pharmaceutics. 2021. PMID: 34834305 Free PMC article. Review.
-
Synthesis, Magnetic Properties, Map Images, and Water Proton Relaxivities of D-Glucuronic Acid Coated Ln2O3 Nanoparticles (Ln = Ho and Er).J Nanosci Nanotechnol. 2015 Sep;15(9):7311-6. doi: 10.1166/jnn.2015.10582. J Nanosci Nanotechnol. 2015. PMID: 26716328
-
Polyethylenimine-Coated Ultrasmall Holmium Oxide Nanoparticles: Synthesis, Characterization, Cytotoxicities, and Water Proton Spin Relaxivities.Nanomaterials (Basel). 2022 May 7;12(9):1588. doi: 10.3390/nano12091588. Nanomaterials (Basel). 2022. PMID: 35564300 Free PMC article.
-
Water-soluble d-glucuronic acid coated ultrasmall mixed Ln/Mn (Ln = Gd and Dy) oxide nanoparticles and their application to magnetic resonance imaging.Biomater Sci. 2014 Sep 29;2(9):1287-1295. doi: 10.1039/c4bm00107a. Epub 2014 Jun 23. Biomater Sci. 2014. PMID: 32481899
-
Ultrasmall Europium, Gadolinium, and Dysprosium Oxide Nanoparticles: Polyol Synthesis, Properties, and Biomedical Imaging Applications.Mini Rev Med Chem. 2020;20(17):1767-1780. doi: 10.2174/1389557520666200604163452. Mini Rev Med Chem. 2020. PMID: 32496986 Review.
Cited by
-
Functionalized Lanthanide Oxide Nanoparticles for Tumor Targeting, Medical Imaging, and Therapy.Pharmaceutics. 2021 Nov 8;13(11):1890. doi: 10.3390/pharmaceutics13111890. Pharmaceutics. 2021. PMID: 34834305 Free PMC article. Review.
-
Magnetic Nanoparticle-Based High-Performance Positive and Negative Magnetic Resonance Imaging Contrast Agents.Pharmaceutics. 2023 Jun 15;15(6):1745. doi: 10.3390/pharmaceutics15061745. Pharmaceutics. 2023. PMID: 37376193 Free PMC article. Review.
-
Potential Applications of Rare Earth Metal Nanoparticles in Biomedicine.Pharmaceuticals (Basel). 2025 Jan 24;18(2):154. doi: 10.3390/ph18020154. Pharmaceuticals (Basel). 2025. PMID: 40005968 Free PMC article. Review.
-
Enhanced Tumor Imaging Using Glucosamine-Conjugated Polyacrylic Acid-Coated Ultrasmall Gadolinium Oxide Nanoparticles in Magnetic Resonance Imaging.Int J Mol Sci. 2022 Feb 4;23(3):1792. doi: 10.3390/ijms23031792. Int J Mol Sci. 2022. PMID: 35163714 Free PMC article.
-
Special Issue "Advanced Nanomaterials for Bioimaging".Nanomaterials (Basel). 2022 Jul 20;12(14):2496. doi: 10.3390/nano12142496. Nanomaterials (Basel). 2022. PMID: 35889719 Free PMC article.
References
-
- Rudin M. Molecular Imaging: Basic Principles and Applications in Biomedical Research. Imperial College Press; London, UK: 2005.
-
- Pankhurst Q., Thanh N.T.K., Jones S.K., Dobson J. Progress in applications of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2009;42:224001. doi: 10.1088/0022-3727/42/22/224001. - DOI
-
- Roca A.G., Costo R., Rebolledo A.F., Veintemillas-Verdaguer S., Tartaj P., Gonzalezcarreno T., Morales M.D.P., Serna C.J. Progress in the preparation of magnetic nanoparticles for applications in biomedicine. J. Phys. D: Appl. Phys. 2009;42:224002. doi: 10.1088/0022-3727/42/22/224002. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

