Partial Agonist Activity of Neonicotinoids on Rat Nicotinic Receptors: Consequences over Epinephrine Secretion and In Vivo Blood Pressure
- PMID: 34065933
- PMCID: PMC8151892
- DOI: 10.3390/ijms22105106
Partial Agonist Activity of Neonicotinoids on Rat Nicotinic Receptors: Consequences over Epinephrine Secretion and In Vivo Blood Pressure
Abstract
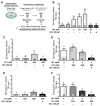
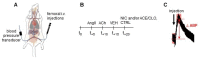
Neonicotinoid insecticides are nicotine-derived molecules which exert acute neurotoxic effects over the insect central nervous system by activating nicotinic acetylcholine receptors (nAChRs). However, these receptors are also present in the mammalian central and peripheral nervous system, where the effects of neonicotinoids are faintly known. In mammals, cholinergic synapses are crucial for the control of vascular tone, blood pressure and skeletal muscle contraction. We therefore hypothesized that neonicotinoids could affect cholinergic networks in mammals and sought to highlight functional consequences of acute intoxication in rats with sub-lethal concentrations of the highly used acetamiprid (ACE) and clothianidin (CLO). In this view, we characterized their electrophysiological effects on rat α3β4 nAChRs, knowing that it is predominantly expressed in ganglia of the vegetative nervous system and the adrenal medulla, which initiates catecholamine secretion. Both molecules exhibited a weak agonist effect on α3β4 receptors. Accordingly, their influence on epinephrine secretion from rat adrenal glands was also weak at 100 μM, but it was stronger at 500 μM. Challenging ACE or CLO together with nicotine (NIC) ended up with paradoxical effects on secretion. In addition, we measured the rat arterial blood pressure (ABP) in vivo by arterial catheterization. As expected, NIC induced a significant increase in ABP. ACE and CLO did not affect the ABP in the same conditions. However, simultaneous exposure of rats to both NIC and ACE/CLO promoted an increase of ABP and induced a biphasic response. Modeling the interaction of ACE or CLO on α3β4 nAChR is consistent with a binding site located in the agonist pocket of the receptor. We present a transversal experimental approach of mammal intoxication with neonicotinoids at different scales, including in vitro, ex vivo, in vivo and in silico. It paves the way of the acute and chronic toxicity for this class of insecticides on mammalian organisms.
Keywords: acetamiprid; acute intoxication; blood pressure; clothianidin; epinephrine secretion; neonicotinoids; nicotine; α3β4 nAChR.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Clothianidin, a neonicotinoid insecticide, activates α4β2, α7 and muscarinic receptors to induce in vivo dopamine release from rat striatum.Toxicology. 2019 Oct 1;426:152285. doi: 10.1016/j.tox.2019.152285. Epub 2019 Aug 31. Toxicology. 2019. PMID: 31479693
-
Injection of insect membrane in Xenopus oocyte: An original method for the pharmacological characterization of neonicotinoid insecticides.J Pharmacol Toxicol Methods. 2016 Jan-Feb;77:10-6. doi: 10.1016/j.vascn.2015.09.004. Epub 2015 Sep 25. J Pharmacol Toxicol Methods. 2016. PMID: 26391340
-
Imidacloprid, a neonicotinoid insecticide, facilitates tyrosine hydroxylase transcription and phenylethanolamine N-methyltransferase mRNA expression to enhance catecholamine synthesis and its nicotine-evoked elevation in PC12D cells.Toxicology. 2018 Feb 1;394:84-92. doi: 10.1016/j.tox.2017.12.004. Epub 2017 Dec 12. Toxicology. 2018. PMID: 29246838
-
A critical review of neonicotinoid insecticides for developmental neurotoxicity.Crit Rev Toxicol. 2016 Feb;46(2):153-90. doi: 10.3109/10408444.2015.1090948. Epub 2015 Oct 29. Crit Rev Toxicol. 2016. PMID: 26513508 Free PMC article. Review.
-
Selective toxicity of neonicotinoids attributable to specificity of insect and mammalian nicotinic receptors.Annu Rev Entomol. 2003;48:339-64. doi: 10.1146/annurev.ento.48.091801.112731. Epub 2002 Jun 4. Annu Rev Entomol. 2003. PMID: 12208819 Review.
Cited by
-
Thiacloprid Induced Developmental Neurotoxicity via ROS-Oxidative Injury and Inflammation in Chicken Embryo: The Possible Attenuating Role of Chicoric and Rosmarinic Acids.Biology (Basel). 2021 Oct 25;10(11):1100. doi: 10.3390/biology10111100. Biology (Basel). 2021. PMID: 34827094 Free PMC article.
-
Neurotoxic Effects of Neonicotinoids on Mammals: What Is There beyond the Activation of Nicotinic Acetylcholine Receptors?-A Systematic Review.Int J Mol Sci. 2021 Aug 5;22(16):8413. doi: 10.3390/ijms22168413. Int J Mol Sci. 2021. PMID: 34445117 Free PMC article.
-
A lethal dose of insight: A case report of acetamiprid and fipronil self-poisoning.Toxicol Rep. 2025 Feb 24;14:101968. doi: 10.1016/j.toxrep.2025.101968. eCollection 2025 Jun. Toxicol Rep. 2025. PMID: 40115001 Free PMC article.
-
HCN2-Associated Neurodevelopmental Disorders: Data from Patients and Xenopus Cell Models.Ann Neurol. 2025 Sep;98(3):573-589. doi: 10.1002/ana.27277. Epub 2025 Jun 5. Ann Neurol. 2025. PMID: 40468825 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

