Newborn Screening and Treatment of Phenylketonuria: Projected Health Outcomes and Cost-Effectiveness
- PMID: 34065950
- PMCID: PMC8151371
- DOI: 10.3390/children8050381
Newborn Screening and Treatment of Phenylketonuria: Projected Health Outcomes and Cost-Effectiveness
Abstract
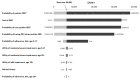
The objective of this study was to evaluate the cost-effectiveness of newborn screening and treatment for phenylketonuria (PKU) in the context of new data on adherence to recommended diet treatment and a newly available drug treatment (sapropterin dihydrochloride). A computer simulation model was developed to project outcomes for a hypothetical cohort of newborns with PKU. Four strategies were compared: (1) clinical identification (CI) with diet treatment; (2) newborn screening (NBS) with diet treatment; (3) CI with diet and medication (sapropterin dihydrochloride); and (4) NBS with diet and medication. Data sources included published literature, primary data, and expert opinion. From a societal perspective, newborn screening with diet treatment had an incremental cost-effectiveness ratio of $6400/QALY compared to clinical identification with diet treatment. Adding medication to NBS with diet treatment resulted in an incremental cost-effectiveness ratio of more than $16,000,000/QALY. Uncertainty analyses did not substantially alter the cost-effectiveness results. Newborn screening for PKU with diet treatment yields a cost-effectiveness ratio lower than many other recommended childhood prevention programs even if adherence is lower than previously assumed. Adding medication yields cost-effectiveness results unlikely to be considered favorable. Future research should consider conditions under which sapropterin dihydrochloride would be more economically attractive.
Keywords: cost-effectiveness; newborn screening; phenylketonuria; sapropterin dihydrochloride.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Kleyn M., Audruszewski K. Michigan Newborn Screening Program, Annual Report 2015. Michigan Department of Health and Human Services; Hart, MI, USA: 2017.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

