Prospects of Using Biocatalysis for the Synthesis and Modification of Polymers
- PMID: 34067052
- PMCID: PMC8124709
- DOI: 10.3390/molecules26092750
Prospects of Using Biocatalysis for the Synthesis and Modification of Polymers
Abstract
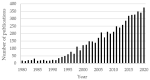
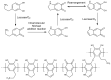
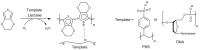
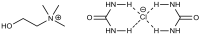
Trends in the dynamically developing application of biocatalysis for the synthesis and modification of polymers over the past 5 years are considered, with an emphasis on the production of biodegradable, biocompatible and functional polymeric materials oriented to medical applications. The possibilities of using enzymes not only as catalysts for polymerization but also for the preparation of monomers for polymerization or oligomers for block copolymerization are considered. Special attention is paid to the prospects and existing limitations of biocatalytic production of new synthetic biopolymers based on natural compounds and monomers from biomass, which can lead to a huge variety of functional biomaterials. The existing experience and perspectives for the integration of bio- and chemocatalysis in this area are discussed.
Keywords: biobased polymers; biocatalysis; biocatalytic monomer synthesis; biocompatible polymers; biodegradable polymers; enzymatic polymerization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
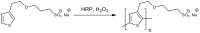










Similar articles
-
Biocatalytic synthesis of conducting polymers and prospects for its application.Biochemistry (Mosc). 2013 Dec;78(13):1539-53. doi: 10.1134/S0006297913130117. Biochemistry (Mosc). 2013. PMID: 24490739 Review.
-
Enzymatic polymerization: Recent advances toward sustainable polymer synthesis.Biotechnol Adv. 2025 Jul-Aug;81:108566. doi: 10.1016/j.biotechadv.2025.108566. Epub 2025 Mar 19. Biotechnol Adv. 2025. PMID: 40118227 Review.
-
Immobilized Candida antarctica lipase B catalyzed synthesis of biodegradable polymers for biomedical applications.Biomater Sci. 2019 Nov 19;7(12):4963-4983. doi: 10.1039/c9bm00716d. Biomater Sci. 2019. PMID: 31532401 Review.
-
Enzyme Catalyst Engineering toward the Integration of Biocatalysis and Chemocatalysis.Trends Biotechnol. 2021 Nov;39(11):1173-1183. doi: 10.1016/j.tibtech.2021.01.002. Epub 2021 Feb 4. Trends Biotechnol. 2021. PMID: 33551176 Review.
-
Synthesis and biomedical applications of functional poly(α-hydroxy acids) via ring-opening polymerization of O-carboxyanhydrides.Acc Chem Res. 2015 Jul 21;48(7):1777-87. doi: 10.1021/ar500455z. Epub 2015 Jun 11. Acc Chem Res. 2015. PMID: 26065588
Cited by
-
Latest Trends in Lipase-Catalyzed Synthesis of Ester Carbohydrate Surfactants: From Key Parameters to Opportunities and Future Development.Int J Mol Sci. 2024 Mar 27;25(7):3727. doi: 10.3390/ijms25073727. Int J Mol Sci. 2024. PMID: 38612540 Free PMC article. Review.
-
Synthesis of Novel Polymer-Assisted Organic-Inorganic Hybrid Nanoflowers and Their Application in Cascade Biocatalysis.Molecules. 2023 Jan 14;28(2):839. doi: 10.3390/molecules28020839. Molecules. 2023. PMID: 36677897 Free PMC article.
-
Polymerization potential of a bacterial CotA-laccase for β-naphthol: enzyme structure and comprehensive polymer characterization.Front Microbiol. 2024 Nov 21;15:1501112. doi: 10.3389/fmicb.2024.1501112. eCollection 2024. Front Microbiol. 2024. PMID: 39640860 Free PMC article.
-
Biocatalysis: A smart and green tool for the preparation of chiral drugs.Chirality. 2022 Nov;34(11):1403-1418. doi: 10.1002/chir.23498. Epub 2022 Aug 5. Chirality. 2022. PMID: 35929567 Free PMC article. Review.
-
Ring-Opening Polymerization of Trimethylene Carbonate with Phosphazene Organocatalyst.Polymers (Basel). 2023 Jan 31;15(3):720. doi: 10.3390/polym15030720. Polymers (Basel). 2023. PMID: 36772021 Free PMC article.
References
-
- Torrelo G., Hanefeld U., Hollmann F. Biocatalysis. Catal. Lett. 2015;145:309–345. doi: 10.1007/s10562-014-1450-y. - DOI
-
- Li G., Zhang H., Sun Z., Liu X., Reetz M.T. Multiparameter optimization in directed evolution: Engineering thermostability, enantioselectivity, and activity of an epoxide hydrolase. ACS Catal. 2016;6:3679–3687. doi: 10.1021/acscatal.6b01113. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

