A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study
- PMID: 34067248
- PMCID: PMC8224682
- DOI: 10.3390/nu13061763
A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study
Abstract
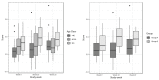
Personalized nutrition may be more effective in changing lifestyle behaviors compared to population-based guidelines. This single-arm exploratory study evaluated the impact of a 10-week personalized systems nutrition (PSN) program on lifestyle behavior and health outcomes. Healthy men and women (n = 82) completed the trial. Individuals were grouped into seven diet types, for which phenotypic, genotypic and behavioral data were used to generate personalized recommendations. Behavior change guidance was also provided. The intervention reduced the intake of calories (-256.2 kcal; p < 0.0001), carbohydrates (-22.1 g; p < 0.0039), sugar (-13.0 g; p < 0.0001), total fat (-17.3 g; p < 0.0001), saturated fat (-5.9 g; p = 0.0003) and PUFA (-2.5 g; p = 0.0065). Additionally, BMI (-0.6 kg/m2; p < 0.0001), body fat (-1.2%; p = 0.0192) and hip circumference (-5.8 cm; p < 0.0001) were decreased after the intervention. In the subgroup with the lowest phenotypic flexibility, a measure of the body's ability to adapt to environmental stressors, LDL (-0.44 mmol/L; p = 0.002) and total cholesterol (-0.49 mmol/L; p < 0.0001) were reduced after the intervention. This study shows that a PSN program in a workforce improves lifestyle habits and reduces body weight, BMI and other health-related outcomes. Health improvement was most pronounced in the compromised phenotypic flexibility subgroup, which indicates that a PSN program may be effective in targeting behavior change in health-compromised target groups.
Keywords: dietary intervention; healthy lifestyle; mixed meal tolerance test; personalized nutrition; systems biology.
Conflict of interest statement
I.M. de Hoogh, S. Bijlsma, T. Krone, T.J. van den Broek, M.P.M. Caspers and S. Wopereis are employees of the Netherlands Organization for Applied Scientific Research (TNO), a not-for-profit research organization collaborating in several public–private partnerships or business-to-business research projects that receive funding from companies. B.L. Winters received funding from Habit, LLC for the design and implementation of the trial and from TNO for preparation of the methods section of the manuscript. K.M. Nieman and B.D. Anderson received research funding from Habit, LLC for the design and implementation of the trial described. J.C. Anthony was previously Chief Science Officer, an advisor, and held shares in Habit, LLC, which was previously owned by the Campbell Soup Company. JCA remains a shareholder of the Campbell Soup Company. The study described in the current publication was funded by Habit LLC.
Figures

Similar articles
-
Caloric intake and eating behavior in infants and toddlers with cystic fibrosis.Pediatrics. 2002 May;109(5):E75-5. doi: 10.1542/peds.109.5.e75. Pediatrics. 2002. PMID: 11986481
-
[Simple obesity in children. A study on the role of nutritional factors].Med Wieku Rozwoj. 2006 Jan-Mar;10(1):3-191. Med Wieku Rozwoj. 2006. PMID: 16733288 Review. Polish.
-
The Impact of a Weight Loss Intervention on Diet Quality and Eating Behaviours in People with Obesity and COPD.Nutrients. 2017 Oct 20;9(10):1147. doi: 10.3390/nu9101147. Nutrients. 2017. PMID: 29053575 Free PMC article.
-
High-soluble-fiber foods in conjunction with a telephone-based, personalized behavior change support service result in favorable changes in lipids and lifestyles after 7 weeks.J Am Diet Assoc. 2002 Apr;102(4):503-10. doi: 10.1016/s0002-8223(02)90116-1. J Am Diet Assoc. 2002. PMID: 11985406 Clinical Trial.
-
Dietary Guidelines for Breast Cancer Patients: A Critical Review.Adv Nutr. 2017 Jul 14;8(4):613-623. doi: 10.3945/an.116.014423. Print 2017 Jul. Adv Nutr. 2017. PMID: 28710147 Free PMC article. Review.
Cited by
-
Digital Biomarkers for Personalized Nutrition: Predicting Meal Moments and Interstitial Glucose with Non-Invasive, Wearable Technologies.Nutrients. 2022 Oct 24;14(21):4465. doi: 10.3390/nu14214465. Nutrients. 2022. PMID: 36364728 Free PMC article.
-
Research gaps and opportunities in precision nutrition: an NIH workshop report.Am J Clin Nutr. 2022 Dec 19;116(6):1877-1900. doi: 10.1093/ajcn/nqac237. Am J Clin Nutr. 2022. PMID: 36055772 Free PMC article.
-
Personalized optimal nutrition lifestyle for self obesity management using metaalgorithms.Sci Rep. 2022 Jul 20;12(1):12387. doi: 10.1038/s41598-022-16260-w. Sci Rep. 2022. PMID: 35858966 Free PMC article.
-
Chrononutrition: Potential, Challenges, and Application in Managing Obesity.Int J Mol Sci. 2025 May 26;26(11):5116. doi: 10.3390/ijms26115116. Int J Mol Sci. 2025. PMID: 40507926 Free PMC article. Review.
-
The Future of Obesity Management through Precision Nutrition: Putting the Individual at the Center.Curr Nutr Rep. 2024 Sep;13(3):455-477. doi: 10.1007/s13668-024-00550-y. Epub 2024 May 28. Curr Nutr Rep. 2024. PMID: 38806863 Free PMC article. Review.
References
-
- U.S. Department of Health. Human Services. U.S. Department of Agriculture 2015–2020 Dietary Guidelines for Americans. [(accessed on 29 July 2020)]; Available online: https://health.gov/our-work/food-nutrition/2015-2020-dietary-guidelines/...
-
- Yubero-Serrano E.M., Delgado-Lista J., Tierney A.C., Perez-Martinez P., Garcia-Rios A., Alcala-Diaz J.F., Castaño J.P., Tinahones F.J., Drevon C.A., Defoort C., et al. Insulin Resistance Determines a Differential Response to Changes in Dietary Fat Modification on Metabolic Syndrome Risk Factors: The LIPGENE Study. Am. J. Clin. Nutr. 2015;102:1509–1517. doi: 10.3945/ajcn.115.111286. - DOI - PubMed
-
- Kirwan L., Walsh M.C., Celis-Morales C., Marsaux C.F.M., Livingstone K.M., Navas-Carretero S., Fallaize R., O’Donovan C.B., Woolhead C., Forster H., et al. Phenotypic Factors Influencing the Variation in Response of Circulating Cholesterol Level to Personalised Dietary Advice in the Food4Me Study. Br. J. Nutr. 2016;116:2011–2019. doi: 10.1017/S0007114516004256. - DOI - PubMed
-
- Blanco-Rojo R., Alcala-Diaz J.F., Wopereis S., Perez-Martinez P., Quintana-Navarro G.M., Marin C., Ordovas J.M., van Ommen B., Perez-Jimenez F., Delgado-Lista J., et al. The Insulin Resistance Phenotype (Muscle or Liver) Interacts with the Type of Diet to Determine Changes in Disposition Index after 2 Years of Intervention: The CORDIOPREV-DIAB Randomised Clinical Trial. Diabetologia. 2016;59:67–76. doi: 10.1007/s00125-015-3776-4. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

