Nanostructured ZnFe2O4: An Exotic Energy Material
- PMID: 34068267
- PMCID: PMC8153140
- DOI: 10.3390/nano11051286
Nanostructured ZnFe2O4: An Exotic Energy Material
Abstract
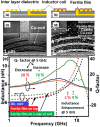
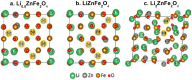
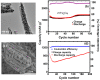
More people, more cities; the energy demand increases in consequence and much of that will rely on next-generation smart materials. Zn-ferrites (ZnFe2O4) are nonconventional ceramic materials on account of their unique properties, such as chemical and thermal stability and the reduced toxicity of Zn over other metals. Furthermore, the remarkable cation inversion behavior in nanostructured ZnFe2O4 extensively cast-off in the high-density magnetic data storage, 5G mobile communication, energy storage devices like Li-ion batteries, supercapacitors, and water splitting for hydrogen production, among others. Here, we review how aforesaid properties can be easily tuned in various ZnFe2O4 nanostructures depending on the choice, amount, and oxidation state of metal ions, the specific features of cation arrangement in the crystal lattice and the processing route used for the fabrication.
Keywords: energy harvesting and storage; inverted ZnFe2O4; nanostructuration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




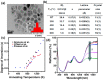
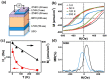
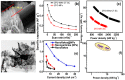
 x = 0.104;
x = 0.104;  x = 0.134;
x = 0.134;  x = 0.159;
x = 0.159;  x = 0.203).
x = 0.203).


References
-
- Dalapati G.K., Chua C.S., Kushwaha A., Liew S.L., Suresh V., Chi D. All earth abundant materials for low cost solar-driven hydrogen production. Mater. Lett. 2016;183:183–186. doi: 10.1016/j.matlet.2016.07.098. - DOI
-
- Abdel Maksoud M.I.A., Fahim R.A., Shalan A.E., Abd Elkodous M., Olojede S.O., Osman A.I., Farrell C., Al-Muhtaseb A.H., Awed A.S., Ashour A.H., et al. Advanced materials and technologies for supercapacitors used in energy conversion and storage: A review. Environ. Chem. Lett. 2021;19:375–439.
-
- Wong H., Iwai H. The road to miniaturization. Phys. World. 2005;18:40–44. doi: 10.1088/2058-7058/18/9/31. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

